PlasmoGEM
Parasites and Microbes
Archive Page
This page is maintained as a historical record and is no longer being updated.
Malaria is a mosquito transmitted disease that still poses a major public health problem to a large portion of the world, with 216 million cases and nearly 0.5 million deaths in 2016. Malaria is caused by Plasmodium parasites that are transmitted to people through the bites of infected female Anopheles mosquitoes. Plasmodium are single cell eukaryotes with complex life cycles, and are only very distantly related to model eukaryotes such as yeast or nematode worms. As a result, almost two decades since the completion of the first Plasmodium reference genome, nearly 50% of the 5,000+ genes in the genome lack an annotated function.
PlasmoGEM (the Plasmodium Genetic Modification Project) is an interdisciplinary group at the Wellcome Sanger Institute focussed on changing the scale of malaria experimental genetics.
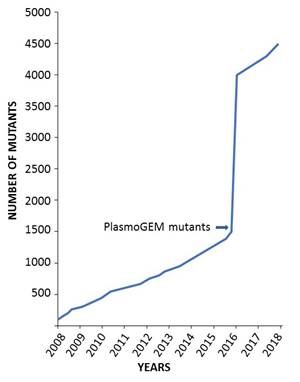
PlasmoGEM develops new genetic tools and makes them freely available to the malaria research community. It also uses those tools to carry out large-scale genetic screens, such as a recent screen that quantified blood-stage growth rates for knockouts of over half the Plasmodium berghei genome (Bushell et al., Cell 2017).
PlasmoGEM is a collaborative project shared across the Rayner, Lee and Billker groups.
Previous core team members

Dr Ana Rita Batista Gomes
Postdoctoral Fellow

Dr Oliver Billker
Former Senior Group Leader

Burcu Bronner-Anar
Technician Commitment Manager

Hannah Bruce
Advanced Research Assistant

Dr Ellen Bushell
Senior Staff Scientist

Mr Gareth Girling
Advanced Research Assistant

Colin Herd
Advanced Research Assistant

Dr Marcus Lee
Former Group Leader

Tom Metcalf
Advanced Research Assistant

Dr Julian C Rayner
Former Senior Group Leader at the Sanger Institute and former Director of Wellcome Connecting Science

Dr Theo Sanderson
Postdoctoral Fellow

Dr Frank Schwach
Principal Bioinformatician

Dr Zenon A Zenonos
Postdoctoral Fellow
