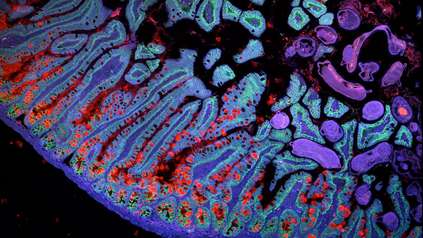
Sanger researchers to tackle deadly brain cancer with ‘tissue time machine’
Researchers at the Sanger Institute are starting work on a Wellcome Leap programme, as part of the ‘Delta Tissue’ or ‘tissue time machine’ project. The project aims to profile the state of cells and tissues – that is their types, developmental trajectories, interactions, features, forms and functions – and predict the transition between states. In collaboration with researchers at the University of Cambridge, the German Cancer Research Centre (DKFZ) and The Francis Crick Institute, the Sanger Institute team will profile Glioblastoma multiforme (GBM) tumours using the latest genomic techniques. Their work will provide unprecedented detail about the states of these cancer cells and their tissue microenvironment, and potentially uncover new targets for treatment development.
Glioblastoma multiforme (GBM) is an aggressive type of brain tumour that is currently incurable. GBM is the most common type of primary malignant brain tumour in adults. The tumours are very varied – they contain a mixture of different cell types, and there is high heterogeneity in the genetic, transcriptional and epigenetic activity of the cells. Recent research has found that different states are highly dynamic in GBM, with cells transitioning from one state to another throughout tumour development as well as in disease relapse after therapy.
To understand the forces that drive the cell changes seen in GBM tumours, Sanger researchers plan to comprehensively characterise the cells and molecules of the tumour tissue in 2D and 3D. This will include using spatial genomic methods that can map the intracellular signals and interactions that influence the tumour cells – something that is not currently well understood. They will initially focus on immune cells and their role in the GBM by profiling samples from 50 patients. The samples will be taken during neurosurgeries by clinicians at Addenbrooke’s Hospital in Cambridge.
“This work will allow us to pinpoint molecular interactions between tumour cells and immune cells as well as interactions with systems in the brain such as the vascular system. We hope that in those interactions we can find a potential treatment target for this devastating disease.”
Professor David Rowitch, Co-Principal Investigator of the study, University of Cambridge
The team will study the cells using a range of techniques. These include single-cell DNA sequencing to determine an individual cell’s genome sequence and any mutations present, and single-cell RNA sequencing to determine which areas of the genome are active within an individual cell. The researchers will also use spatial transcriptomics to uncover precisely which cells within a tissue are using which genes. Data from all of these methods will be integrated to build a comprehensive, unbiased picture of cell states in GBM.
Building on previous work, the team will use computational approaches to spatially map BGM cell types and to identify intracellular signals. They aim to identify molecular signals sent between immune and tumour cells, as well as those involved in neurodevelopmental processes that have a role in GBM.
‘We’re excited about the challenges of integrating the rich multi-omics data modalities generated in this unique initiative. The ability to integrate large-scale multi-modal data will also allow us to go beyond a mere descriptive catalogue to make steps towards causality. In particular the ability to map tumorigenesis in the spatial context of tissues will give us strong pointers to what is actually causing these tumours, and what might be the best type of drug targets for investigation.”
Oliver Stegle, Co-Principal Investigator, DFKZ and Associate Faculty Member, Wellcome Sanger institute
“This is a very different approach to what has been tried before. I think that these technologies give us a decent shot at identifying therapeutically important molecules or cell interactions that is just wasn’t possible to see before. I am incredibly excited to be exploiting large scale data generation and cutting edge computational tools to improve our understanding of this incurable brain tumour.”
Omer Bayraktar, Principal Investigator, Wellcome Sanger Institute
More information
Positions available – related to the Glioblastoma multiforme (GBM) Spatial Multi-omics project:
Computational Postdoctoral Fellow in GBM Spatial Multi-omics:
https://jobs.sanger.ac.uk/vacancy/computational-postdoctoral-fellow-in-gbm-spatial-multiomics-464137.html
Postdoctoral Fellow in GBM Spatial Multi-omics:
https://jobs.sanger.ac.uk/vacancy/postdoctoral-fellow-in-gbm-spatial-multiomics-464139.html
Staff scientist in GBM Spatial Multi-omics:
https://jobs.sanger.ac.uk/vacancy/staff-scientist-in-gbm-spatial-multiomics-464132.html
Advanced Research Assistant in Spatial Genomics:
https://jobs.sanger.ac.uk/vacancy/advanced-research-assistant-in-spatial-genomics-461728.html