1 in 5 patients with breast cancer could benefit from existing treatment, genetic study reveals
Breast cancer is the most common cancer in the UK, affecting nearly 55,000 women a year. Globally it accounts for nearly 1.7 million cancer cases. Between 1 and 5 per cent of breast cancer cases are due to inherited mutations in BRCA1 or BRCA2 genes.
Inherited mutations in BRCA1 and BRCA2 genes mean that some of the machinery required to fix DNA is faulty. People with these faults have higher risks of developing certain cancers, like breast and ovarian cancer.
Drugs called PARP inhibitors have been designed to specifically treat tumours with faulty BRCA1 and BRCA2 genes in breast and ovarian cancers, and their use against prostate cancer is currently being investigated.
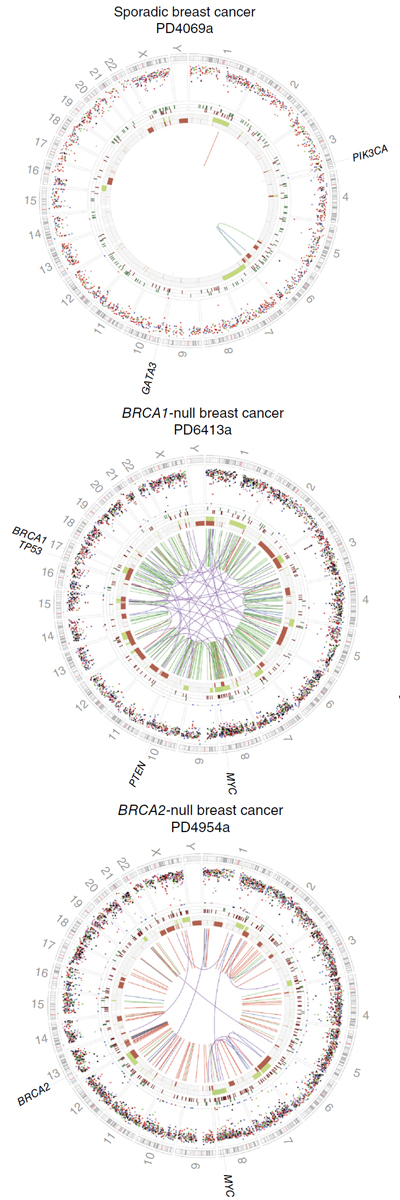
In the study, researchers analysed the breast cancer genomes of 560 patients and looked for every single type of mutation possible. The team developed a new computer-based tool called HRDetect to identify patterns of mutations – mutational signatures – in the tumours, which were similar to people who have mutations in the BRCA1 and BRCA2 genes.
Scientists discovered that many breast cancer patients had mutational signatures that were identical to people with faulty BRCA1 and BRCA2 genes, even though they had not inherited the mutations.
The results suggest that roughly 1 in 5 breast cancer patients could benefit from existing PARP inhibitor treatments. This would need to be tested through a clinical trial, with participants being selected based on the mutational signatures of their tumour.
“In the past, clinical trials for PARP inhibitors have focused mainly on the 1 to 5 per cent of women with breast cancer. However, our study shows that there are many more people who have cancers that look like they have the same signatures and same weakness as patients with faulty BRCA1 and BRCA2 genes. We should explore if they could also benefit from PARP inhibitors. The results suggest that clinical trials now need to look at cancer patients who share the same genetic signature in their cancer. This could change how clinical trials are designed in the future.”
Dr Serena Nik-Zainal Lead author from the Wellcome Trust Sanger Institute
Until now, most clinical trials of PARP inhibitors have focused on patients with inherited mutations in the BRCA1 and BRCA2 genes. This research suggests that systematic clinical trials on a wider set of patients will be required to see if they might also be responsive to the drugs.
“From the mutational signatures we were able to spot many more tumours with defects in their DNA repair machinery that we couldn’t see before. This was only possible by sequencing the entire genome of these cancers. Further work needs to be done as there could be tumours with the same mutational signature elsewhere in the body that may respond to these drugs.”
Dr Helen Davies Joint first author from the Sanger Institute
“This work uses mutational signatures to identify the complete set of cancers that will respond to certain drugs that are already known to be effective in a subset. To translate these results into treatments, further sequencing of cancer genomes and more clinical trials are urgently needed, but this is a most promising start.”
Professor Sir Mike Stratton Director of the Sanger Institute
More information
What are PARP inhibitors?
PARP inhibitors are a group of pharmacological inhibitors of the enzyme poly ADP ribose polymerase (PARP). Several forms of cancer are more dependent on PARP for DNA repair than regular cells, making it an attractive target for cancer therapy. For cancers that are BRCA1/BRCA2-deficient, PARP is an alternative DNA repair mechanism that the cancer cells rely on. The drugs work by blocking the PARP DNA repair mechanism in BRCA1/BRCA2-deficient cancer cells, so damaged DNA is not mended, leading to cancer cell death.
What is HRDetect?
HRDetect is a new tool in the form of a computer programme developed by the authors of this study. It is able to discriminate between tumours with and without the BRCA1/BRCA2 mutation, also known as a deficiency in homologous recombination (HR), based on the appearance of their genome. Cancer genomes without a mutation in the BRCA1 and BRCA2 genes are neat and organised, whereas genomes with BRCA1/BRCA2-deficiency are chaotic. HRDetect sees the tumour profile as if it is reading an x-ray, and can be applied across all tumours.
Funding:
This work was supported by the European Community’s Seventh Framework Programme, the Wellcome Trust, the Institut National du Cancer (inca), France and the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea.
Publications:
Selected websites
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world's leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
Wellcome
Wellcome exists to improve health for everyone by helping great ideas to thrive. We’re a global charitable foundation, both politically and financially independent. We support scientists and researchers, take on big problems, fuel imaginations and spark debate.