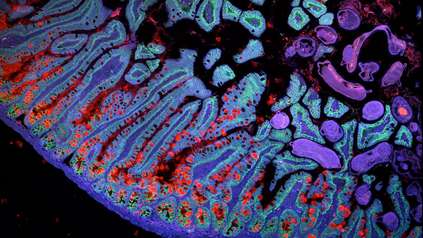
Study identifies trigger for ‘head-to-tail’ axis development in human embryo
Scientists have identified key molecular events in the developing human embryo between days 7 and 14, giving greater understanding of critical stages of our development.
The research, a collaboration including scientists from the Wellcome Sanger Institute and the University of Cambridge, gives new information on how these cells develop and what molecular and cellular mechanisms are involved.
In this study, published 17th June 2021 in Nature Communications, researchers created a cell atlas of the human embryo by using embryos cultured in vitro to see the changes that they go through pre and post implantation, shedding new light on human development.
The second week of gestation represents a critical stage of embryo development, or embryogenesis. Failure of development during this time is one of the major causes of early pregnancy loss. Understanding more about it will help scientists to understand how it can go wrong, and take steps towards being able to fix problems.
The pre-implantation period, before the developing embryo implants into the mother’s womb, has been studied extensively in human embryos in the lab. On the seventh day the embryo must implant into the womb to survive and develop. Very little is known about the development of the human embryo once it implants, because it becomes inaccessible for study.
This study used embryos cultured in vitro (outside of the body) both before and after implantation but pre gestation (where the embryo matures fully), allowing them to be studied up to day 14 of development for the first time.
In this research, scientists from the Wellcome Sanger Institute and the University of Cambridge studied what happens at the molecular level during this early stage of embryogenesis. Their findings provide the first evidence that a group of cells outside the embryo, known as the hypoblast, send a message to the embryo that initiates the development of the head-to-tail body axis.
When the body axis begins to form, the symmetrical structure of the embryo starts to change. One end becomes committed to developing into the head end, and the other the ‘tail’.
By using single-cell sequencing methodologies and functional experiments, the research revealed molecular signals involved in the formation of the body axis show similarities to those in animals, despite significant differences in the positioning and organisation of the cells.
“We have revealed the patterns of gene expression in the developing embryo just after it implants in the womb, which reflect the multiple conversations going on between different cell types as the embryo develops through these early stages. Our goal has always been to enable insights to very early human embryo development in a dish, to understand how our lives start. By combining our new technology with advanced sequencing methods we have delved deeper into the key changes that take place at this incredible stage of human development, when so many pregnancies unfortunately fail.”
Professor Magdalena Zernicka-Goetz, senior author and Professor of Mammalian Development and Stem Cell Biology at the University of Cambridge
The study identified the gene conversations in the developing embryo by sequencing the code in the thousands of messenger RNA molecules made by individual cells. They captured the evolving molecular profile of the developing embryo after implantation in the womb, revealing the progressive loss of pluripotency (the ability of the embryonic cells to give rise to any cell type of the future organism) as the fates of different cells are determined.
“By creating an atlas of the cells involved in human development and how they communicate with other cells, we can start to understand more about the cellular processes and mechanisms behind very early embryo growth, which has been much harder to study compared to other mammals. This freely available information can now be used by researchers around the world to help inform future studies.”
Dr Roser Vento-Tormo, senior author and Group Leader at the Wellcome Sanger Institute
“The early days of embryo development are critical, yet there is little known about the processes involved. By applying the latest in genomic sequencing, imaging and culture techniques we are able to shed some light on what happens in these early stages. Hopefully in the future, this can be used to understand the extent of how interactions in early human development impact later life.”
Dr Sam Behjati, senior author and Group Leader and Wellcome Trust Intermediate Clinical Fellow at the Wellcome Sanger Institute
More information
This study carried out with the oversight of the UK Human Fertilisation and Embryology Authority, and with permission from a local research ethics committee.
Publication:
Mole, M.A. et al. ‘A single cell characterisation of human embryogenesis identifies pluripotency transitions and putative anterior hypoblast centre.’ June 2021. Nature Communications. DOI: 10.1038/s41467-021-23758-w
Funding:
This research was funded by Wellcome, the European Molecular Biology Organisation, UKRI Medical Research Council, the Gates Cambridge Trust, Open Philanthropy/Silicon Valley, Curci and Weston Havens Foundations. For full funding information, please see the publication.