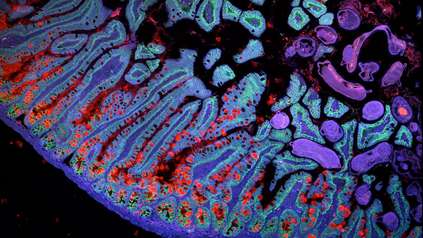
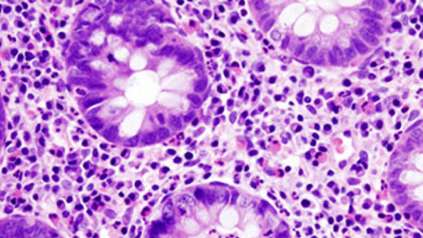
Gut research identifies key cellular changes associated with childhood-onset Crohn’s Disease
Scientists have tracked the very early stages of human foetal gut development in incredible detail, and found specific cell functions that appear to be reactivated in the gut of children with Crohn’s Disease. The results are an important step towards better management and treatment of this devastating condition.
The research from the University of Cambridge and the Wellcome Sanger Institute is part of the global Human Cell Atlas initiative to map every cell type in the human body. The findings reveal intricate cellular mechanisms of how the gut develops.
Crohn’s Disease is a type of Inflammatory Bowel Disease affecting around one in every 650 people in the UK. Incidence has increased dramatically in recent decades, especially in children – who can suffer very aggressive symptoms including abdominal pain, diarrhoea and fatigue. This lifelong condition can have major life implications; the cause is not understood, treatments often don’t work, and there is no cure.
“Crohn’s Disease can be particularly aggressive and more treatment-resistant in children, so there’s a real need to understand the condition when it affects them and perhaps come up with childhood-specific treatments.”
Dr Matthias Zilbauerlead author on the study from the Department of Paediatrics at the University of Cambridge and honorary consultant in paediatric gastroenterology at Addenbrooke’s Hospital*
The researchers used a cutting-edge technology called single-cell RNA sequencing to look at gene expression in individual cells of the developing human gut, six to ten weeks after conception. They focused on the inner lining of the gut, called the intestinal epithelium, and found that the cells there divide constantly at this early stage, guided by messages from other cell types. This allows the gut to grow, and form the structures needed for good gut function later in life.
Tissues from the guts of children with Crohn’s Disease, aged between four and twelve, were also analysed. The study revealed that some of the cellular pathways active in the epithelium of the foetal gut appear to be reactivated in Crohn’s Disease. These pathways were not active in healthy children of a similar age. The results are published today in the journal Developmental Cell.
“Our results indicate there might be a reprogramming of specific gut cell functions in Crohn’s Disease. We don’t know whether this is the cause of the disease or a consequence of it, but either way it is an exciting step in helping us to better understand the condition.”
Dr Matthias Zilbauer
The findings shed light on fundamental molecular mechanisms of human gut development. The team also found that lab-grown ‘mini-guts’ undergo similar individual cellular changes as those inside a developing foetus. This implies that lab-grown models are a powerful and accurate tool for future research into very early gut development and associated diseases.
“The gut starts as a simple tube and develops into a highly complex organ, involved in food digestion, immunity and even control of feeding behaviour. In our study we were able to take snapshots of cell types during development, and work out the molecular interactions that guide the gut epithelial cells to change from a simple tube, to complex structures forming finger-like protrusions.”
Rasa Elmentaite, a first author on the study from the Wellcome Sanger Institute
“This study is part of the international Human Cell Atlas effort to create a ‘Google map’ of the entire human body. With single-cell RNA sequencing we can look at any tissue and identify the individual cell types it’s made up of, the function of those cells, and even identify new cell types.
“A complex tissue like the gut contains different cell types, and these ‘talk’ to each other – the function of one cell affects the function of another. That’s particularly important in the early stages of gut development, and something we can interrogate using computational analyses of single cell RNA sequencing data.”
Dr Sarah Teichmann, a senior author from the Wellcome Sanger Institute, and co-chair of the Human Cell Atlas Organising Committee
While the study focused specifically on the dynamics of intestinal epithelial cells, it generated information on around 90,000 primary human intestinal cells of all types. The researchers have made this data openly available at www.gutcellatlas.org, creating a valuable resource for further research and drug discovery targeted at childhood Crohn’s Disease.
“From my own experience we’re diagnosing Crohn’s Disease in younger and younger children, some even under the age of five – it’s very much an emerging disease. It’s a really nasty, lifelong condition, and when children are diagnosed, the whole family is affected. We are determined to advance our knowledge in this area, and hopefully improve the lives of these children in the future.”
Dr Matthias Zilbauer
More information
Publication:
Elmentaite, R. and Ross, A. et al. (2020) Single-cell sequencing of developing human gut reveals transcriptional links to childhood Crohn’s disease. Developmental Cell. DOI: 10.1016/j.devcel.2020.11.010
https://www.cell.com/developmental-cell/fulltext/S1534-5807(20)30886-8
*Addenbrooke’s Hospital in Cambridge is a specialist centre for the investigation and treatment of Inflammatory Bowel Disease in children, serving the east of England.
Funding:
This research was supported by the Medical Research Council, Wellcome, the Great Ormond Street Hospital Children’s Charity, Sparks, and other funders. Please see the paper for all funding information.
Selected websites
About the University of Cambridge
The mission of the University of Cambridge is to contribute to society through the pursuit of education, learning and research at the highest international levels of excellence. To date, 110 affiliates of the University have won the Nobel Prize.
Founded in 1209, the University comprises 31 autonomous Colleges, which admit undergraduates and provide small-group tuition, and 150 departments, faculties and institutions. Cambridge is a global university. Its 19,000 student body includes 3,700 international students from 120 countries. Cambridge researchers collaborate with colleagues worldwide, and the University has established larger-scale partnerships in Asia, Africa and America.
The University sits at the heart of the ‘Cambridge cluster’, which employs 60,000 people and has in excess of £12 billion in turnover generated annually by the 4,700 knowledge-intensive firms in and around the city. The city publishes 341 patents per 100,000 residents.
About the Wellcome Sanger Institute
The Wellcome Sanger Institute is a world leading genomics research centre. We undertake large-scale research that forms the foundations of knowledge in biology and medicine. We are open and collaborative; our data, results, tools and technologies are shared across the globe to advance science. Our ambition is vast – we take on projects that are not possible anywhere else. We use the power of genome sequencing to understand and harness the information in DNA. Funded by Wellcome, we have the freedom and support to push the boundaries of genomics. Our findings are used to improve health and to understand life on Earth. Find out more at www.sanger.ac.uk or follow us on Twitter, Facebook, LinkedIn and on our Blog.
About Wellcome
Wellcome exists to improve health by helping great ideas to thrive. We support researchers, we take on big health challenges, we campaign for better science, and we help everyone get involved with science and health research. We are a politically and financially independent foundation. https://wellcome.org