New research on brain structure highlights cells linked to Alzheimer’s and autism
New insights into the architecture of the brain have been revealed by scientists at the Wellcome Sanger Institute, the Wellcome-MRC Cambridge Stem Cell Institute and their collaborators. The researchers discovered that cells in the cerebral cortex of mice, called astrocytes, are more diverse than previously thought, with distinct layers of astrocytes across the cerebral cortex that provide the strongest evidence to date of their specialization across the brain.
Published today (16 March) in Nature Neuroscience, the most in-depth study of its kind is set to change the way we think about the brain and the role of cells such as astrocytes. This knowledge will have implications for the study of neurological disorders, such as Alzheimer’s, multiple sclerosis and autism.
In the past 20 years, research has shown glial cells to be key players in brain development and function, as well as promising targets for better understanding neurological disorders. Alzheimer’s causes around two thirds of dementia cases in the UK, which affects around 850,000 individuals at present*. MS is a neurological disorder that affects the central nervous system and impacts around 100,000 people in the UK**. Autism affects around one in every hundred people in the UK***.
‘Glial’ comes from the Greek word for ‘glue’ or ‘putty’. At one time, glial cells were thought of as ‘brain putty’ – functionally similar, passive cells whose only function was to fill the space around the ‘all important’ neurons. However, new studies are showing their critical importance in regulating neuron functions^. Astrocytes are a type of glial cell, so called because of their ‘star-shaped’ structure^^.
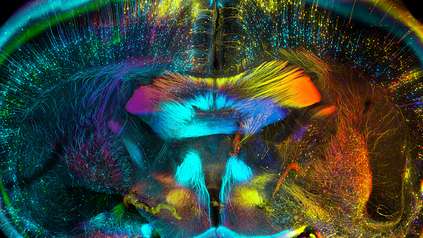
Despite the wealth of knowledge on neuronal function and the organisation of neurons into layers, prior to this study there had been little investigation into whether glial cells across different layers showed different cellular properties. To answer this question, the researchers developed a new methodological approach to provide a more detailed view of the organisation of astrocytes than ever before.
Nucleic acid imaging was carried out on mouse and human brain samples at the University of Cambridge to map how new genes are expressed within tissue. These maps were combined with single cell genomic data at the Wellcome Sanger Institute to extend the molecular description of astrocytes. These data sets were then combined to create a three-dimensional, high-resolution picture of astrocytes in the cerebral cortex.
The team discovered that astrocytes are not uniform as previously thought, but take distinct molecular forms depending on their location in the cerebral cortex. They found that astrocytes are also organised into multiple layers, but that the boundaries of astrocyte layers are not identical to the neuronal layers. Instead, astrocyte layers have less sharply defined edges and overlap the neuronal layers.
“The discovery that astrocytes are organised into layers that are similar, but not identical to, neuronal layers redefines our view of the structure of the mammalian brain. The structure of the cerebral cortex can no longer simply be seen as the structure of neurons. If you want to properly understand how our brains work, you have to consider how astrocytes are organised and what role they play.”
Dr Omer Bayraktar, Group Leader at the Wellcome Sanger Institute
As well as increasing our understanding of brain biology, the findings will have implications for the study and treatment of human neurological disorders. Over the past decade glial cells, rather than neurons, have been heavily implicated in diseases such as Alzheimer’s and multiple sclerosis.
“This study shows that the cortical architecture is more complex than previously thought. It provides a basis to begin to understand the precise roles played by astrocytes, and how they are involved in human neurodevelopmental and neurodegenerative diseases.”
Professor David Rowitch, Head of Paediatrics at the University of Cambridge
More information
In the cerebral cortex of the mammalian brain, neurons are the cells responsible for transmitting information throughout the body. It has long been recognised that the 10-14 billion neurons of the human cerebral cortex are organised into six layers, with distinct populations of neurons in each layer that correspond to their function https://www.dartmouth.edu/~rswenson/NeuroSci/chapter_11.html
* More information on Alzheimer’s disease can be found here: https://www.alzheimersresearchuk.org/about-dementia/types-of-dementia/alzheimers-disease/about/
**More information about MS can be found here: https://www.mssociety.org.uk/about-ms/what-is-ms
*** More Information on autism is available from the National Autistic Society: https://www.autism.org.uk/about/what-is/asd.aspx
^ An overview of the changing status of glial cells is available at: https://blogs.scientificamerican.com/brainwaves/know-your-neurons-meet-the-glia/
^^ Only half of the cells in the human cerebral cortex are neurons, the other half are glial cells, of which astrocytes are a type. The molecular signals that astrocytes provide are essential for forming synapses between neurons. They regulate synapse formation in the developing brain, as well as refining synapses in the maturing brain – ‘pruning’ extra synapses to ‘sculpt’ neuronal networks.
Publication:
Omer Ali Bayraktar, Theresa Bartels and Staffan Holmqvist et al. (2020). Astrocyte layers in the mammalian cerebral cortex revealed by a single-cell in situ transcriptomic map. Nature Neuroscience. https://doi.org/10.1038/s41593-020-0602-1
Funding:
The study was supported by the Dr Miriam and Sheldon G. Adelson Medical Research Foundation, National Institute of Health (1R01 MH109912; P01NS08351), NINDS Informatics Center for Neurogenetics and Neurogenomics (P30 NS062691), Wellcome and the European Research Council (281961).
Selected websites
Wellcome - MRC Cambridge Stem Cell Institute
The Wellcome – MRC Cambridge Stem Cell Institute is a world-leading centre for stem cell research with a mission to transform human health through a deep understanding of normal and pathological stem cell behaviour. Bringing together biological, clinical and physical scientists operating across a range of tissue types and at multiple scales, we explore the commonalities and differences in stem cell biology in a cohesive and inter-disciplinary manner. In 2019, we relocated to a new purpose-built home on the Cambridge Biomedical Campus. Housing over 350 researchers, including a critical mass of clinician scientists, the Institute integrates with neighbouring disease-focused research institutes and also serves as a hub for the wider stem cell community in Cambridge.
About the University of Cambridge
The mission of the University of Cambridge is to contribute to society through the pursuit of education, learning and research at the highest international levels of excellence. To date, 107 affiliates of the University have won the Nobel Prize. Founded in 1209, the University comprises 31 autonomous Colleges, which admit undergraduates and provide small-group tuition, and 150 departments, faculties and institutions. Cambridge is a global university. Its 19,000 student body includes 3,700 international students from 120 countries. Cambridge researchers collaborate with colleagues worldwide, and the University has established larger-scale partnerships in Asia, Africa and America. The University sits at the heart of the ‘Cambridge cluster’, which employs 60,000 people and has in excess of £12 billion in turnover generated annually by the 4,700 knowledge-intensive firms in and around the city. The city publishes 341 patents per 100,000 residents. www.cam.ac.uk
The Wellcome Sanger Institute
The Wellcome Sanger Institute is a world leading genomics research centre. We undertake large-scale research that forms the foundations of knowledge in biology and medicine. We are open and collaborative; our data, results, tools and technologies are shared across the globe to advance science. Our ambition is vast – we take on projects that are not possible anywhere else. We use the power of genome sequencing to understand and harness the information in DNA. Funded by Wellcome, we have the freedom and support to push the boundaries of genomics. Our findings are used to improve health and to understand life on Earth. Find out more at www.sanger.ac.uk or follow us on Twitter, Facebook, LinkedIn and on our Blog.
About Wellcome
Wellcome exists to improve health by helping great ideas to thrive. We support researchers, we take on big health challenges, we campaign for better science, and we help everyone get involved with science and health research. We are a politically and financially independent foundation. https://wellcome.org/