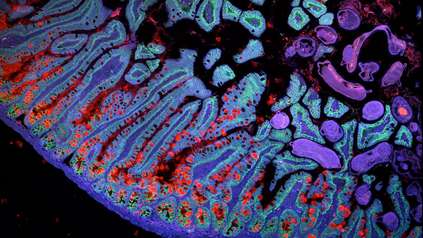
First spatial map of human limb development reveals unexpected growth processes and explains syndromes found at birth
Human fingers and toes do not grow outward; instead, they form from within a larger foundational bud, as intervening cells recede to reveal the digits beneath. This is among many processes captured for the first time as scientists unveil a spatial cell atlas of the entire developing human limb, resolved in space and time.
In this latest study, which is part of the Human Cell Atlas initiative to map every cell type in the human body1, researchers at the Wellcome Sanger Institute, Sun Yat-sen University, EMBL’s European Bioinformatics Institute and collaborators, applied cutting-edge single-cell and spatial technologies to create an atlas characterising the cellular landscape and pinpointing the exact location of cells of the early human limb.
The atlas, published today (6 December) in Nature, provides an openly available resource that captures the intricate processes governing the limbs’ rapid development during the early stages of limb formation2.
The atlas also uncovers new links between developmental cells and some congenital limb syndromes, such as short fingers and extra digits.
Limbs are known to initially emerge as undifferentiated cell pouches on the sides of the body, without a specific shape or function. But after 8 weeks of development, they are well differentiated, anatomically complex and immediately recognisable as limbs, complete with fingers and toes. This requires a very rapid and precise orchestration of cells. Any small disturbances to this process can have a downstream effect, which is why variations in the limbs are among the most frequently reported syndromes at birth, affecting approximately one in 500 births globally3.
While limb development has been extensively studied in mouse and chick models, the extent to which they mirror human situation remained unclear. However, advances in technology now enable researchers to explore the early stages of human limb formation.
In this new study, scientists from the Wellcome Sanger Institute, Sun Yat-sen University, and their collaborators analysed tissues between 5 and 9 weeks of development. This allowed them to trace specific gene expression programs, activated at certain times and in specific areas, which shape the forming limbs.
Special staining of the tissue revealed clearly how cell populations differentially arrange themselves into patterns of the forming digits4.
As part of the study, researchers demonstrated that certain gene patterns have implications for how the hands and feet form, identifying certain genes, which when disrupted, are associated with specific limb syndromes like brachydactyly – short fingers – and polysyndactyly – extra fingers or toes.
The team were also able to confirm that many aspects of limb development are shared between humans and mice.
Overall, these findings not only provide an in-depth characterisation of limb development in humans but also critical insights that could impact the diagnosis and treatment of congenital limb syndromes.
“Decades of studying model organisms established the basis for our understanding of vertebrate limb development. However, characterising this in humans has been elusive until now, and we couldn’t assume the relevance of mouse models for human development. What we reveal is a highly complex and precisely regulated process. It is like watching a sculptor at work, chiselling away at a block of marble to reveal a masterpiece. In this case, nature is the sculptor, and the result is the incredible complexity of our fingers and toes.”
Professor Hongbo Zhang, senior author of the study from Sun Yat-sen University, Guangzhou
“For the first time, we have been able to capture the remarkable process of limb development down to single cell resolution in space and time. Our work in the Human Cell Atlas is deepening our understanding of how anatomically complex structures from, helping us uncover the genetic and cellular processes behind healthy human development, with many implications for research and healthcare. For instance, we discovered novel roles of key genes MSC andPITX1 that may regulate muscle stem cells. This could offer potential for treating muscle-related disorders or injuries.”
Dr Sarah Teichmann, senior author of the study from the Wellcome Sanger Institute, and co-founder of the Human Cell Atlas
More information
Publication
B. Zhang et al. (2023) ‘A human embryonic limb cell atlas resolved in space and time.’ Nature 2023. DOI: 10.1038/s41586-023-00000-0.
Data Sets
These data sets are available for interactive analysis at: https://limb-dev.cellgeni.sanger.ac.uk/
Footnotes
- This study is part of the international Human Cell Atlas (HCA) consortium, which is aiming to map every cell type in the human body as a basis for both understanding human health and for diagnosing, monitoring, and treating disease. An open, scientist-led consortium, the HCA is a collaborative effort of researchers, institutes, and funders worldwide, with more than 3,000 members from 97 countries across the globe. The HCA is likely to impact every aspect of biology and medicine, propelling translational discoveries and applications and ultimately leading to a new era of precision medicine. More information can be found at https://www.humancellatlas.org/
- The researchers analysed human embryonic limb tissues between weeks 5 to 9 post-conception, provided by Addenbrooke’s hospital Cambridge, United Kingdom, the Women and Children’s Medical Centre, Guangzhou, China and INSERM’s HuDeCA Biobank, Paris, France.
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1571054/
- Video depicts gene expression clusters during limb development through spatial transcriptomic profiles and in situ staining of the tissue: This video shows the dynamic gene expression patterns of IRX1, SOX9 and MSX1, critical genes involved in limb formation. Their distinct distribution ensures the ‘chiselling’ process takes place. IRX1, crucial for digit formation, and SOX9, essential for skeletal development, converge into five distinct lengths within the developing limb, while MSX1, associated with undifferentiated cells, occupies the interdigital spaces between these clusters. At approximately week seven of development, molecules responsible for interdigital cell death are activated, leading to the elimination of cells in the intervening spaces. This orchestrated cell death finally unveils the well-defined shapes of fingers or toes.
Video credit
DOI: 10.1038/s41586-023-00000-0.
Funding
This research was supported by Wellcome and the National Key Research and Development Program of China. For full funding acknowledgements, please refer to the publication.