Potentially cancerous cells kept in check by competitive neighbours, study of oesophagus finds
The expansion of ‘mutant’ cells that could lead to cancer is often kept in check by their neighbours, research from the Wellcome Sanger Institute, the University of Cambridge and their collaborators has found. The team discovered that when equally-matched cells in the oesophagus of mice coincided, they acted as a brake on one another’s growth.
The study, published today in Nature Genetics, describes the ‘rules of the game’ of competition between oesophageal cells for the first time. By understanding these rules, the hope is that therapies can be developed to reduce the competitiveness of mutant clone cells that are more likely to become cancerous.
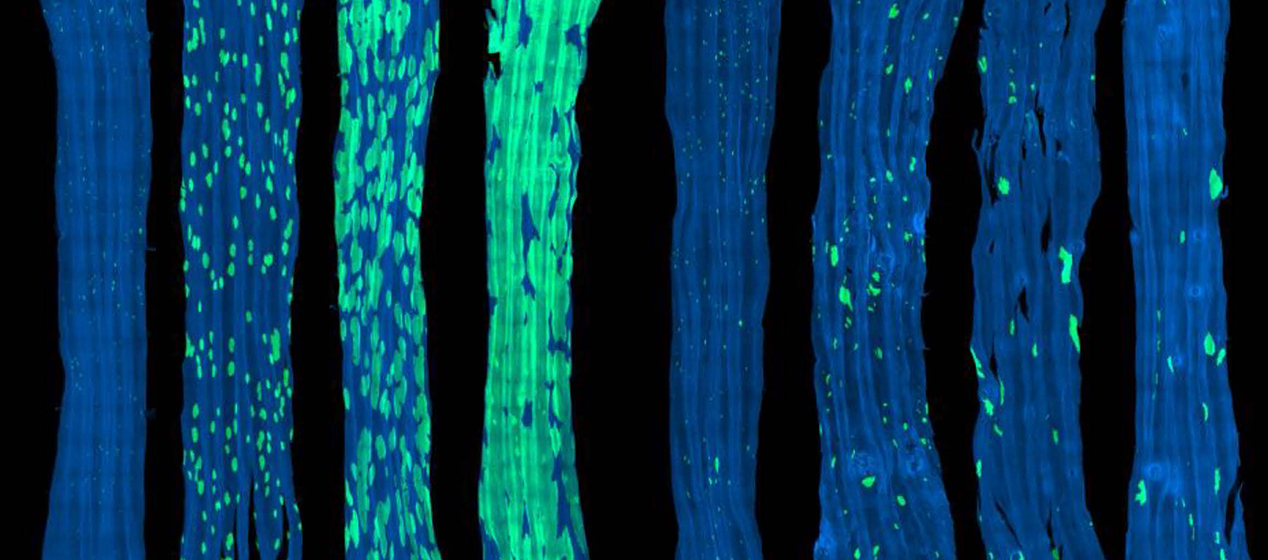
Clones are communities of cells descended from a single stem cell through the normal process of cell division. Tissues in the human body consist of patchworks of clones, with DNA mutations that occur naturally throughout life being passed on from parent to daughter cell.
Some DNA mutations, particularly those affecting known ‘cancer genes’ such as TP53 and NOTCH1, confer a competitive advantage on cells that acquire them. This allows these mutant clones to expand more rapidly than normal cells and can lead to disease.
By middle age, most cells in tissues such as the oesophagus and the skin are mutant clones. But despite this, the vast majority do not go on to form cancers. A previous study* from the Wellcome Sanger Institute has shown that clones competing against each other helps to prevent cancer formation, but until now the ‘rules of the game’ for clonal competition were unknown.
To discover how mutant clones interact, the researchers combined genetic lineage tracing and ultradeep sequencing** to observe the evolving mutational landscape of oesophageal epithelium in mice. A mutagen called diethylnitrosamine – a substance found in tobacco smoke that causes genetic mutations – was administered to generate a patchwork of mutant clones in the mouse to resemble the oesophageal tissue of older humans.
The team found that mutant clone dynamics depend on the mutations they carry and the nature of their neighbouring cells. Expanding mutant clones that bumped up against cells of similar ‘fitness’ lost their competitive advantage and reverted towards the balanced growth of normal tissue.
“When clones acquire certain mutations it’s like they’re playing with loaded dice, while normal cells are still playing with normal dice. This small advantage is enough to help them outcompete other clones and colonise larger areas of tissue over time, which if continued unchecked could lead to disease. In tissues like the oesophagus, which is full of cells carrying cancer-driving mutations, it is actually mutant clones cancelling each other out that helps to keep the tissue healthy.”
Dr Bartomeu Colom, first author of the study from the Wellcome Sanger Institute
“In trying to explain why few mutant clones go on to become cancerous in tissues with high mutational burdens, there has been some debate about whether competition between clones takes place. Our findings show that not only is competition taking place in the oesophageal epithelium, but that it helps to maintain homeostasis and inhibits clones carrying known cancer-driving mutations from continuing down a path towards disease.”
Dr Maria Alcolea, Wellcome-MRC Cambridge Stem Cell Institute
Understanding the dynamics of clonal competition opens up the possibility of designing therapies aimed at reducing the competitive advantage of potentially dangerous clones. By removing these clones or preventing them becoming dominant, the risk of cancer could be reduced.
“In human tissue there is limited space, clones compete for it and the rule of survival of the fittest applies. If we can rig the game against ‘bad’ clones that may go on to form cancer, and in favour of their more benign neighbours, the bad clones will be replaced. By understanding the rules of the game, we can seriously consider how we can intervene and prevent some cancers arising.”
Professor Phil Jones, lead author of the study from the Wellcome Sanger Institute and the MRC Cancer Unit, University of Cambridge
More information
*More information about this study is available here: https://www.sanger.ac.uk/news/view/skin-battlefield-mutations
**Genetic lineage tracing is where single cells are labelled so that their descendants can be tracked under the microscope. Ultradeep sequencing and computational analysis allows identification of the mutations present in small numbers of cells.
Publication:
Bartomeu Colom, Maria P. Alcolea and Gabriel Piedrafita et al. (2020). Spatial competition shapes the dynamic mutational landscape of normal esophageal epithelium. Nature Genetics. DOI: https://doi.org/10.1038/s41588-020-0624-3
Funding:
This research was supported by Wellcome and Cancer Research UK.
Selected websites
Wellcome - MRC Cambridge Stem Cell Institute
The Wellcome – MRC Cambridge Stem Cell Institute is a world-leading centre for stem cell research with a mission to transform human health through a deep understanding of normal and pathological stem cell behaviour. Bringing together biological, clinical and physical scientists operating across a range of tissue types and at multiple scales, we explore the commonalities and differences in stem cell biology in a cohesive and inter-disciplinary manner. In 2019, we relocated to a new purpose-built home on the Cambridge Biomedical Campus. Housing over 350 researchers, including a critical mass of clinician scientists, the Institute integrates with neighbouring disease-focused research institutes and also serves as a hub for the wider stem cell community in Cambridge. https://www.stemcells.cam.ac.uk/
About the University of Cambridge
The mission of the University of Cambridge is to contribute to society through the pursuit of education, learning and research at the highest international levels of excellence. To date, 107 affiliates of the University have won the Nobel Prize.
Founded in 1209, the University comprises 31 autonomous Colleges, which admit undergraduates and provide small-group tuition, and 150 departments, faculties and institutions. Cambridge is a global university. Its 19,000 student body includes 3,700 international students from 120 countries. Cambridge researchers collaborate with colleagues worldwide, and the University has established larger-scale partnerships in Asia, Africa and America.
The University sits at the heart of the ‘Cambridge cluster’, which employs 60,000 people and has in excess of £12 billion in turnover generated annually by the 4,700 knowledge-intensive firms in and around the city. The city publishes 341 patents per 100,000 residents. www.cam.ac.uk
The Wellcome Sanger Institute
The Wellcome Sanger Institute is a world leading genomics research centre. We undertake large-scale research that forms the foundations of knowledge in biology and medicine. We are open and collaborative; our data, results, tools and technologies are shared across the globe to advance science. Our ambition is vast – we take on projects that are not possible anywhere else. We use the power of genome sequencing to understand and harness the information in DNA. Funded by Wellcome, we have the freedom and support to push the boundaries of genomics. Our findings are used to improve health and to understand life on Earth. Find out more at www.sanger.ac.uk or follow us on Twitter, Facebook, LinkedIn and on our Blog.
About Wellcome
Wellcome exists to improve health by helping great ideas to thrive. We support researchers, we take on big health challenges, we campaign for better science, and we help everyone get involved with science and health research. We are a politically and financially independent foundation. https://wellcome.org/