Babies' gut bacteria affected by delivery method, Baby Biome project shows
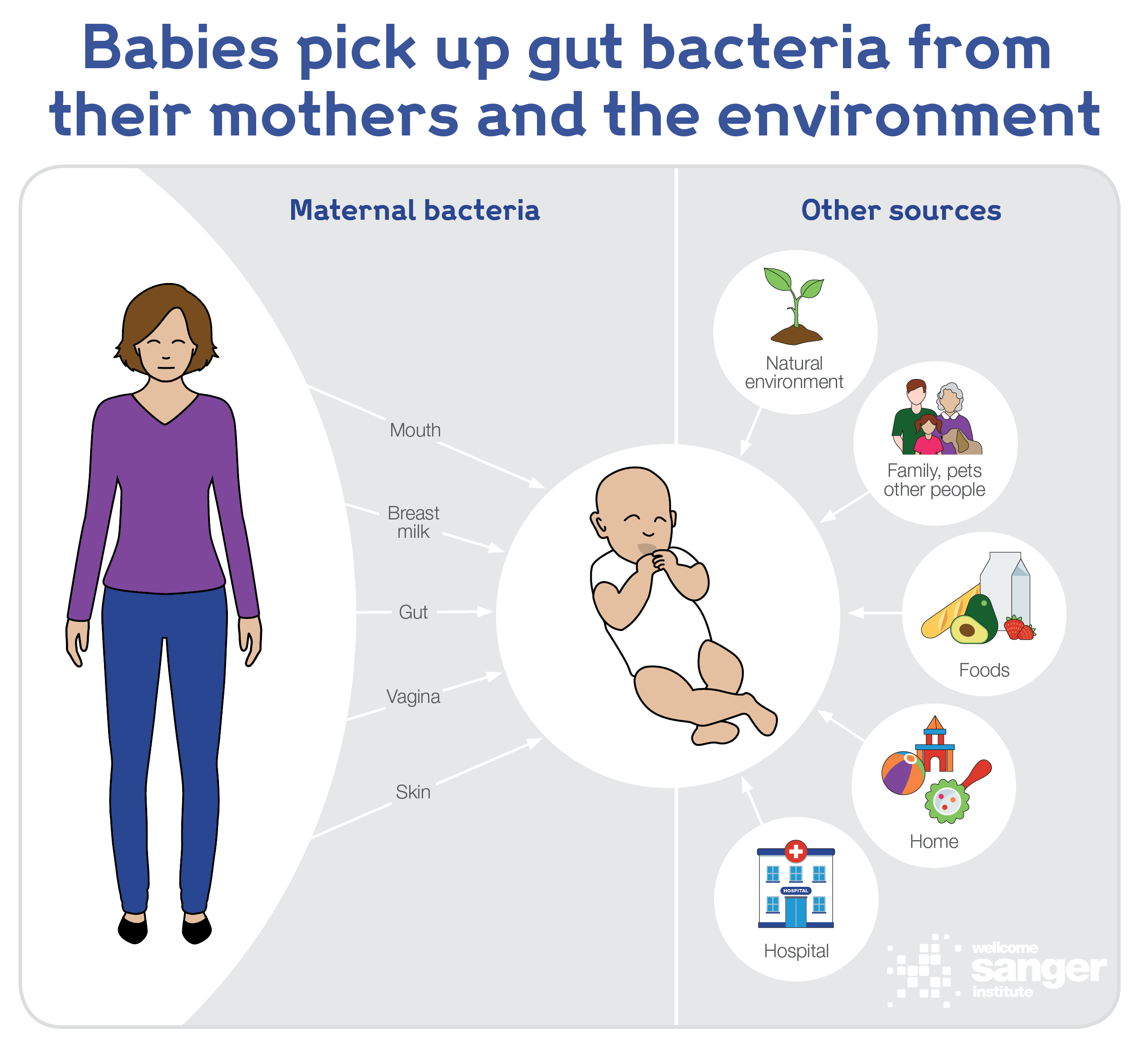
Babies born vaginally have different gut bacteria – their microbiome – than those delivered by Caesarean, research has shown. Scientists from the Wellcome Sanger Institute, UCL, the University of Birmingham and their collaborators discovered that whereas vaginally born babies got most of their gut bacteria from their mother, babies born via caesarean did not, and instead had more bacteria associated with hospital environments in their guts.
Published in Nature today (18th Sept), this largest ever study of neonatal microbiomes also revealed that the microbiome of vaginally delivered newborns did not come from the mother’s vaginal bacteria, but from the mother’s gut. This calls into question the controversial practice of swabbing babies born via caesarean with mother’s vaginal bacteria. Understanding how the birth process impacts on the baby’s microbiome will enable future research into bacterial therapies.
The gut microbiome is a complex ecosystem of millions of microbes, and is thought to be important for the development of the immune system. Lack of exposure to the right microbes in early childhood has been implicated in autoimmune diseases such as asthma, allergies and diabetes. However, it is not fully understood how important the initial gut microbiome is to the baby’s immune system development and health, how a baby’s microbiome develops, or what happens to it with different modes of birth.
To understand more about the development of the microbiome, and if the delivery method affected this, researchers studied 1,679 samples of gut bacteria from nearly 600 healthy babies and 175 mothers. Fecal samples were taken from babies aged four, seven or 21 days old, who had been born in UK hospitals by vaginal delivery or caesarean. Some babies were also followed up later, up to one year of age.
Using DNA sequencing and genomics analysis, the researchers could see which bacteria were present and found there was a significant difference between the two delivery methods. They discovered that vaginally delivered babies had many more health-associated (commensal) bacteria from their mothers, than babies who were born by caesarean.
“This is the largest genomic investigation of newborn babies’ microbiomes to date. We discovered that the mode of delivery had a great impact on the gut bacteria of newborn babies, with transmission of bacteria from mother to baby occurring during vaginal birth. Further understanding of which species of bacteria help create a healthy baby microbiome could enable us to create bacterial therapies.”
Dr Trevor Lawley A senior author on the paper from the Wellcome Sanger Institute
Previous limited studies had suggested that vaginal bacteria were swallowed by the baby on its way down the birth canal. However, this large-scale study found babies had very few of their mother’s vaginal bacteria in their guts, with no difference between babies born vaginally or by caesarean.
During birth, the baby will come into contact with bacteria from the mother’s gut. The study discovered it was the mother’s gut bacteria that made up much of the microbiome in the vaginally delivered babies. Babies born via caesarean had many fewer of these bacteria. This study therefore found no evidence to support controversial ‘vaginal swabbing’ practices, which could transfer dangerous bacteria to the baby.
In place of some of the mother’s bacteria, the babies born via caesarean had more bacteria that are typically acquired in hospitals, and were more likely to have antimicrobial resistance. The researchers isolated, grew and sequenced the genomes of more than 800 of these potentially pathogenic bacteria, confirming that they were the same as strains causing bloodstream infections in UK hospitals. Although these bacteria don’t usually cause disease while in the gut, they can cause infections if they get into the wrong place or if the immune system fails.
“Our study showed that as the babies grow and take in bacteria when they feed and from everything around them, their gut microbiomes become more similar to each other. After they have been weaned, the microbiome differences between babies born via caesarean and delivered vaginally have mainly evened out. We don’t yet know whether the initial differences we found will have any health implications.”
Dr Nigel Field A senior author on the paper from UCL
“In many cases, a Caesarean is a life-saving procedure, and can be the right choice for a woman and her baby. The exact role of the microbiome in the newborn and what factors can change it are still uncertain, so we don’t think this study should deter women from having a caesarean. This study shows that more research is required to improve our understanding of this important area.”
Dr Alison Wright Consultant Obstetrician and Vice President of The Royal College of Obstetricians and Gynaecologists
All women who have a caesarean are now offered antibiotics before the delivery to help prevent the mother developing postoperative infections, meaning that the baby also receives a dose of antibiotics via the placenta. This could also cause some of the microbiome differences seen between the two birth methods.
“The first weeks of life are a critical window of development of the baby’s immune system, but we know very little about it. We urgently need to follow up this study, looking at these babies as they grow to see if early differences in the microbiome lead to any health issues. Further studies will help us understand the role of gut bacteria in early life and could help us develop therapeutics to create a healthy microbiome.”
Professor Peter Brocklehurst University of Birmingham and Principal Investigator of the Baby Biome Study
More information
Publication:
Yan Shao et al. (2019) Stunted gut microbiota and increased pathogen colonisation associated with caesarean birth. Nature. DOI: 10.1038/s41586-019-1560-1
Funding:
This work was supported by Wellcome and the Wellcome Sanger Institute, the Australian National Health and Medical Research Council, and the Victorian Government’s Operational Infrastructure Support Programme.
Selected websites
About UCL
UCL was founded in 1826. We were the first English university established after Oxford and Cambridge, the first to open up university education to those previously excluded from it, and the first to provide systematic teaching of law, architecture and medicine.
We are among the world’s top universities, as reflected by performance in a range of international rankings and tables, and are committed to changing the world for the better.
Our community of over 41,500 students from 150 countries and over 12,500 staff pursues academic excellence, breaks boundaries and makes a positive impact on real world problems.
www.ucl.ac.uk | Follow us on Twitter @uclnews | Watch our YouTube channel YouTube.com/UCLTV
University of Birmingham
The University of Birmingham is ranked amongst the world’s top 100 institutions. Its work brings people from across the world to Birmingham, including researchers, teachers and more than 6,500 international students from over 150 countries. https://www.birmingham.ac.uk
The Wellcome Sanger Institute
The Wellcome Sanger Institute is a world leading genomics research centre. We undertake large-scale research that forms the foundations of knowledge in biology and medicine. We are open and collaborative; our data, results, tools and technologies are shared across the globe to advance science. Our ambition is vast – we take on projects that are not possible anywhere else. We use the power of genome sequencing to understand and harness the information in DNA. Funded by Wellcome, we have the freedom and support to push the boundaries of genomics. Our findings are used to improve health and to understand life on Earth. Find out more at www.sanger.ac.uk or follow us on Twitter, Facebook, LinkedIn and on our Blog.
About Wellcome
Wellcome exists to improve health by helping great ideas to thrive. We support researchers, we take on big health challenges, we campaign for better science, and we help everyone get involved with science and health research. We are a politically and financially independent foundation.