PlasmoGEM - TOOLS
PlasmoGEM is a non-profit, open-access malaria research resource, providing tools for the manipulation of Plasmodium genomes, and using them to carry out large-scale research projects.
PlasmoGEM is a non-profit, open-access malaria research resource, providing tools for the manipulation of Plasmodium genomes, and using them to carry out large-scale research projects. These tools currently include:
P. berghei
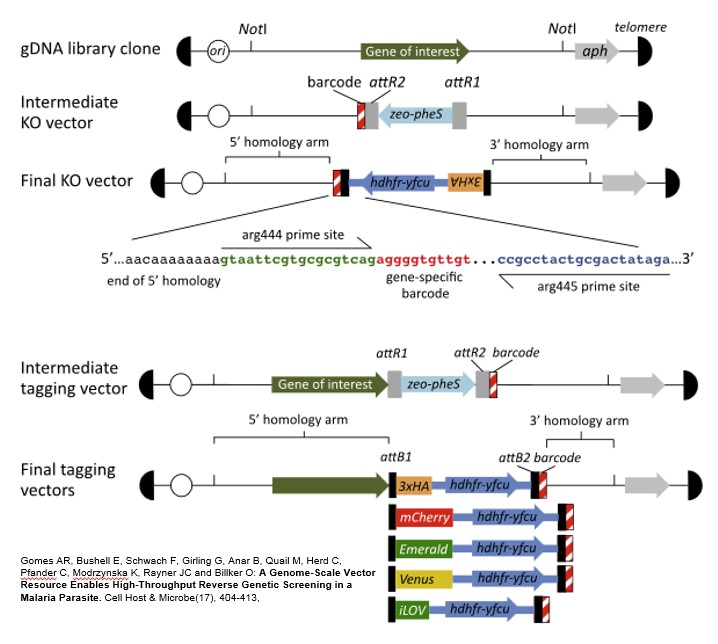
We have produced large numbers of DNA vectors for the targeted manipulation of P. berghei genes. Resources available currently include:
- A large-insert (6-10kb) P. berghei genomic library covering >90% of P. berghei genes
- A library of artificial chromosomes covering >3500 P. berghei genes for use in complementation or over-expression studies.
- Gene targeting vectors for the disruption of >2600 P. berghei genes and epitope tagging of >400 genes. We are currently expanding our knockout library to attempt to cover the entire genome, excluding large multi-gene families such as birs.
- Blood-stage growth phenotypes for >2,600 genes, generated using barcode sequencing after pooled transfection with our gene targeting knock out vectors
All the tools as well as phenotypic data currently available can be found here.
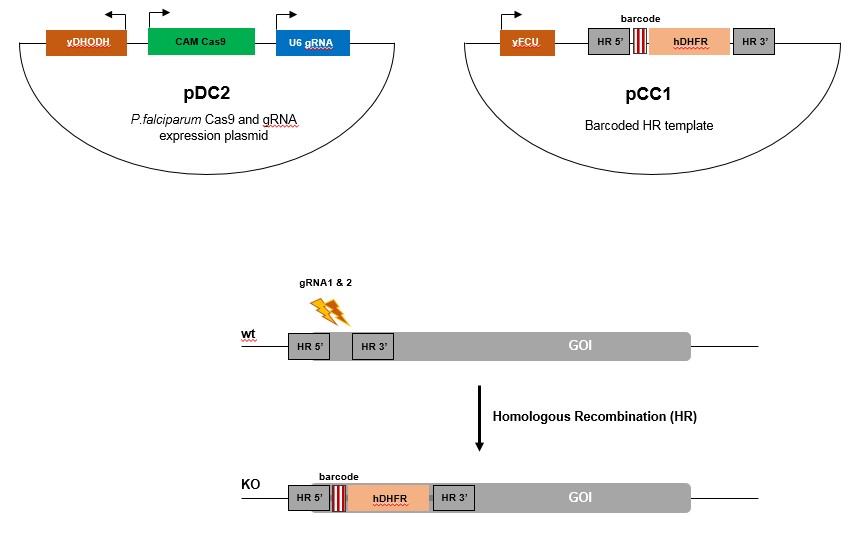
P. falciparum:
PlasmoGEM is currently generating vectors for Cas9 and gRNA expression (pDC2) as well as barcoded homologous repair (HR) templates (pCC1) for 200-300 P.falciparum genes.
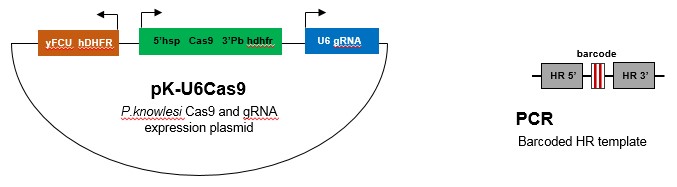
P. knowlesi:
PlasmoGEM is currently generating vectors for Cas9 and gRNA expression (pK-U6Cas9) as well as barcoded homologous repair (HR) templates by PCR for 400-500 P. knowlesi genes.