Vulnerabilities of leukaemia cells revealed using genome editing technique
AML is an aggressive cancer. The cancer cells crowd out healthy cells in the bone marrow. They multiply quickly and interfere with the bone marrow’s ability to make normal blood cells, leading to life-threatening infections and bleeding. Mainstream AML treatments haven’t changed for decades and fewer than one in three people survive the cancer.
To identify new ways to treat AML, the team used CRISPR-Cas9 gene-editing technology to screen cancer cells for vulnerable points. This technology can be used to disrupt and destroy targeted genes in the genomes of cells. To achieve their goals, the team refined a CRISPR-Cas9 technique to efficiently disrupt all genes in the leukaemia cell genome individually. This allowed them to identify those genes whose disruption was detrimental to the growth and survival of AML cells.
“Previous studies showed proof of principle, but this is one of the first systematic attempts to identify the genetic vulnerabilities of AML. We have improved and applied CRISPR-Cas 9 technology to look at what actually kills cells. CRISPR is becoming a powerful technique in cancer research because it overcomes some of the limitations of earlier tools.”
Dr Kosuke Yusa Joint project leader from the Sanger Institute
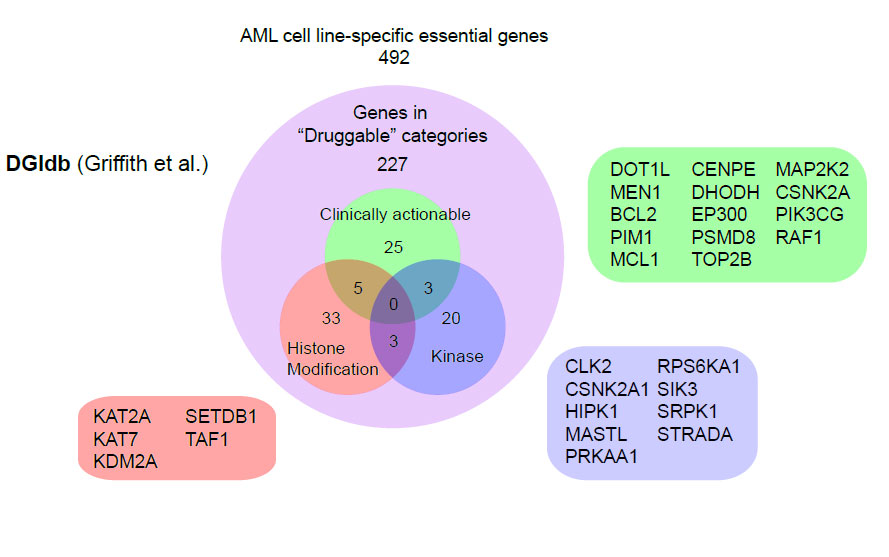
The human genome contains around 20,000 genes, by refining CRISPR-Cas9 technology and using it to screen the leukaemia genome the team uncovered a catalogue of approximately 500 genes that are essential for cancer cell survival, including more than 200 genes for which drugs could be designed. Whilst a handful of these genes including DOT1L, BCL2 and MEN1 are already established therapeutic targets, most of them are novel and open up many new possibilities for developing effective treatments against the disease.
The researchers chose the KAT2A gene for further research and in order to demonstrate the validity of their findings. By inhibiting KAT2A using genetic and drug-based techniques, they showed that disruption of the gene reduced the growth and survival of AML cells, but not of normal blood cells.
“This is an exciting finding, as KAT2A inhibition worked on a number of primary AML cells with diverse genotypes. Whilst the gene needs to be studied in greater depth to understand its potential for use in the clinic, we show that targeting KAT2A destroyed AML cells in the laboratory while sparing healthy blood cells.”
Dr Konstantinos Tzelepis A first author on the paper from the Sanger Institute
The team then validated this finding, by disrupting the KAT2A gene from leukaemia cells in transgenic mice, and observing the effect on the cancer. They found that the mice lived longer when the KAT2A gene was disrupted.
“This research has led to the identification of many potential gene targets for future AML therapy, which we are making available to other researchers to explore. Whilst KAT2A inhibition now needs to be investigated as a treatment strategy for acute myeloid leukaemia, there are many more candidates to pursue by the leukaemia research community. Our hope is that this work will lead to more effective treatments against AML that will improve both the survival and the quality of life of patients.”
Dr George Vassiliou Joint project leader from the Sanger Institute and Consultant Haematologist at Cambridge University Hospitals NHS Trust
More information
Funding
This work was supported by the Kay Kendall Leukaemia Fund (KKLF) and Wellcome.
Publications:
Selected websites
The Kay Kendall Leukaemia Fund
The Kay Kendall Leukaemia Fund was established in 1984 under the Will of the late James Sainsbury CBE. It awards grants for research on aspects of leukaemia and for relevant studies on related haematological malignancies. Grants are awarded for first class research on innovative proposals, particularly those close to the care of leukaemia patients or the prevention of leukaemia or related diseases. Project grants are awarded twice yearly, and Intermediate, and Junior Fellowships of 3-4 years are awarded annually. The Fund also considers support for capital projects that will have direct benefit to leukaemia patient care. For more information please contact the Kay Kendall Leukaemia Fund.
Department of Haematology, University of Cambridge
The University of Cambridge Department of Haematology is located at the Cambridge Biomedical Campus and the Wellcome Trust Genome Campus. It has laboratories in the Cambridge Institute for Medical Research, NHS National Blood and Transplant, and the Sanger Institute. Headed by Professor Tony Green, the department has four main goals: i. To conduct internationally competitive biomedical research, ii. To provide education in medical aspects of haematology to undergraduate scientists and medical students, iii. To provide postgraduate education, largely through the provision of PhD students and iv. To contribute to the clinical activities of the Cambridge University Hospitals Department of Haematology.
Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
Wellcome
Wellcome exists to improve health for everyone by helping great ideas to thrive. We’re a global charitable foundation, both politically and financially independent. We support scientists and researchers, take on big problems, fuel imaginations and spark debate.


