New genetic links for heart disease risk factors identified
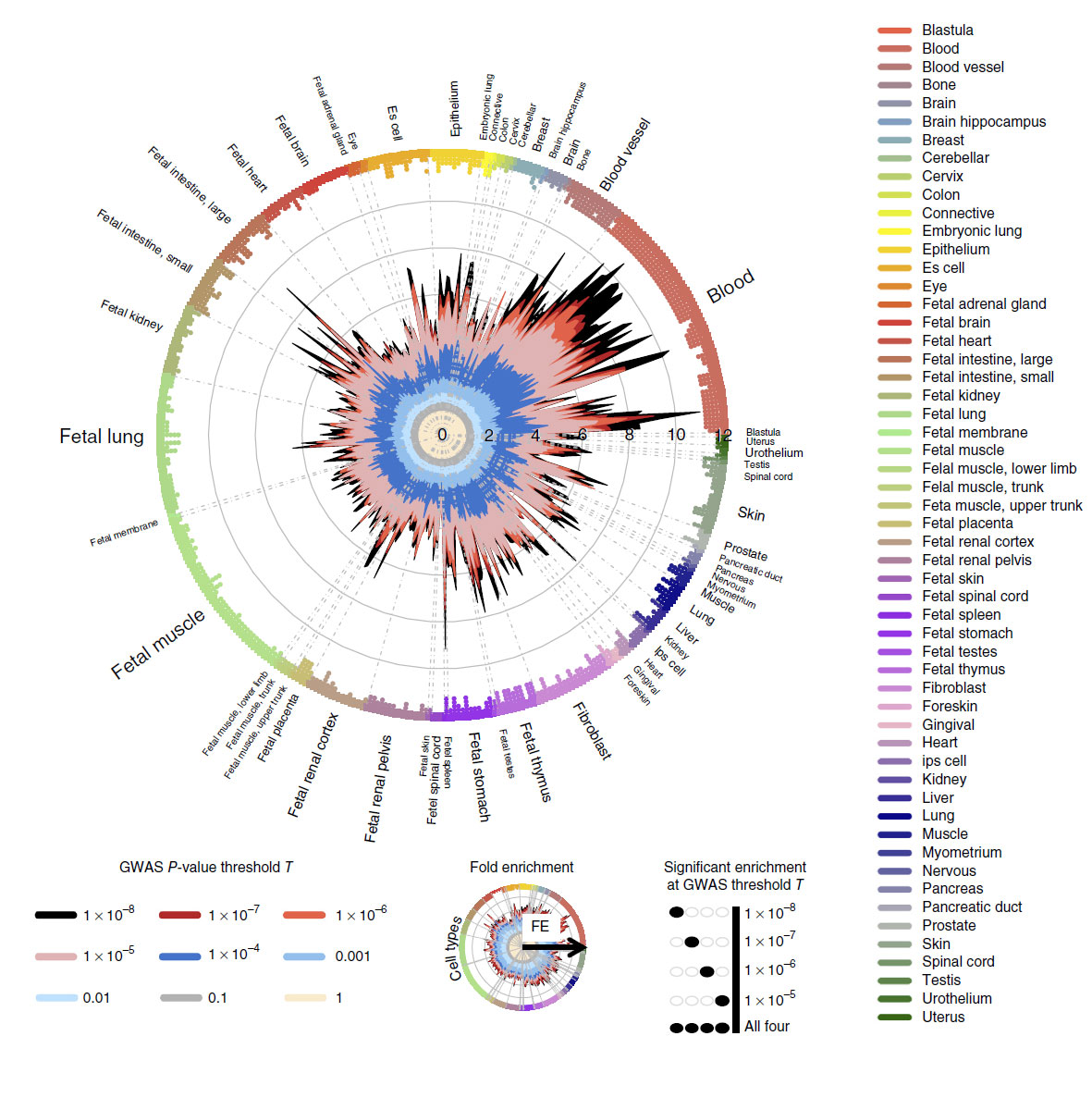
Reported today in Nature Genetics, the research shows how large-scale genomic datasets can be used to help identify potential novel biological targets for studying cardiovascular and other diseases.
Genetics have been implicated in cardiovascular and blood diseases for some time, however as these are complex diseases, it is extremely difficult to find specific genetic causes. In this study, scientists studied the genomes of almost 36,000 healthy people with European ancestry, looking for rare genetic links to 20 known risk factors for disease, such as raised levels of cholesterol or haemoglobin in the blood.
Two previous large-scale projects provided the whole genome sequences needed: the UK10K project – a study of the genetic code of 10,000 people that aims to better understand links between rare genetic variations and disease; and the 1000 genome project. From this data, the scientists created a resource called a dense imputation panel, which is freely accessible to the scientific community. The panel holds so much detail that it can fill in the gaps or ‘impute’ data missing from lower resolution genetic studies.
The level of detail the imputation panel provides enabled the scientists to look at specific disease risk factors, and find 17 new genetic variants. Of these, 16 would have been extremely difficult to find without the imputation panel data.
“The dense imputation panel used in this study allowed us to search for genetic variations that are much less frequent than ever before, but that individually explain a greater genetic risk. As efforts continue to characterise the genetic underpinnings of complex diseases, the methods we have developed in this study are expected to enable the next wave of discoveries of what causes these diseases, and how we might develop new treatments.”
Professor Nicole Soranzo Joint senior author from the Wellcome Trust Sanger Institute
“This is the first stage of a discovery process that is going to tell us more about the contribution genetics makes to complex human diseases. We looked at where in the genome these 17 new variants lie to see what that could tell us about biology; about the changes they make to the body and how that could make a person more or less susceptible to disease.”
Dr Valentina Iotchkova First author from the Sanger Institute and the European Bioinformatics Institute
The researchers then applied an analytical technique called fine-mapping to study hundreds of regions of the human genome that contain genetic risk factors for cardiometabolic disease. For 59 regions, they were able to narrow down the most likely genetic causes to small sets of genetic variants. Combining this fine mapping technique with biological data drilled it down even further and provided additional functional insight into the underlying biology.
“Our study provides a refined method to use the panel alongside other analysis techniques to find rare variants that contribute to complex genetic conditions like heart disease or diabetes. We have also narrowed down the set of potentially causal variants to a small set of variants for further follow up. We focused on cardiometabolic traits but the panel covers the entire human genome so it could be used to glean a deeper understanding of genetic contributions to disease in any part of the human body.”
Dr Paul Auer Joint senior author on the paper from the University of Wisconsin-Milwaukee
More information
Funding
The research was funded by the Wellcome Trust, the European Union Framework Programme 7 and the National Institute for Health Research Blood and Transplant Research Unit. A full list of grant support and can be found in the paper.
Publications:
Selected websites
University of Wisconsin-Milwaukee (UWM)
Recognized as one of the nation’s 115 top research universities, UW-Milwaukee provides a world-class education to more than 27,000 students from 81 countries. Its 14 schools and colleges include Wisconsin’s only schools of architecture, freshwater sciences and public health, and it is a leading educator of nurses and teachers. With a budget of $667 million, UW-Milwaukee partners with leading companies to conduct joint research, offer student internships and serve as an economic engine for southeastern Wisconsin. The Princeton Review named UW-Milwaukee a 2016 “Best Midwestern” university based on overall academic excellence and student reviews.
The European Bioinformatics Institute (EBI)
The European Bioinformatics Institute is part of EMBL, and is a global leader in the storage, analysis and dissemination of large biological datasets. EMBL-EBI helps scientists realise the potential of ‘big data’ by enhancing their ability to exploit complex information to make discoveries that benefit mankind. We are a non-profit, intergovernmental organisation funded by EMBL’s 21 member states and two associate member states. Our 570 staff hail from 57 countries, and we welcome a regular stream of visiting scientists throughout the year. We are located on the Wellcome Genome Campus in Hinxton, Cambridge in the United Kingdom.
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
Wellcome
Wellcome exists to improve health for everyone by helping great ideas to thrive. We’re a global charitable foundation, both politically and financially independent. We support scientists and researchers, take on big problems, fuel imaginations and spark debate.