Single cells seen in unprecedented detail
The study, published today in Nature Methods, has experimentally established for the first time that when a cell loses or gains a copy of a chromosome during cell division, the genes in that particular region of DNA show decreased or increased expression. While this has long been assumed by genetic researchers, it has not been seen before.
The DNA code that makes up our genome can be found in every cell of our body. This is decoded into a group of molecules in our cells known as RNA, which provide instructions for that particular cell’s proteins to perform their particular function. Transcriptome sequencing measures the amount of each RNA molecule in a cell and provides an insight into its function that cannot be gained by looking at the DNA.
Scientists can now see a cell’s DNA, including the mutational damage it has sustained on its journey from the fertilised egg, alongside the RNA of the same cell, which enacts all the DNA’s instructions, even the errors.
“By looking at the genome and transcriptome in parallel in a single cell, we can begin to see what the functional consequences of genetic variations are. This technology will enable us to probe for genetic heterogeneity in normal and diseased tissues deeper than ever before providing novel understanding of normal and disease development; for instance it will help us to better understand the diversity of cancer cells that arise within a tumour, and the difficulty of treating the disease.”
Professor Thierry Voet A corresponding author from KU Leuven and the Wellcome Trust Sanger Institute
G&T-seq is unique in the field as it allows high throughput DNA-and RNA-sequencing from single cells in parallel on a diverse range of sequencers, while previous methods were limited to the interrogation of either the DNA or the RNA of a cell, but not both.
“The potential to scale-up this method is one of the most exciting elements of this research. To meaningfully grasp the heterogeneity of cells in a tissue, we need to be able to look at genome and transcriptome data for hundreds or thousands of individual cells at a time. As the cost of sequencing continues to drop, these large-scale projects will become more feasible and we will begin to see, in unprecedented detail, the diversity and life-history of cells that form human tissue.”
Dr Iain Macaulay First author and a corresponding author from the Sanger Institute
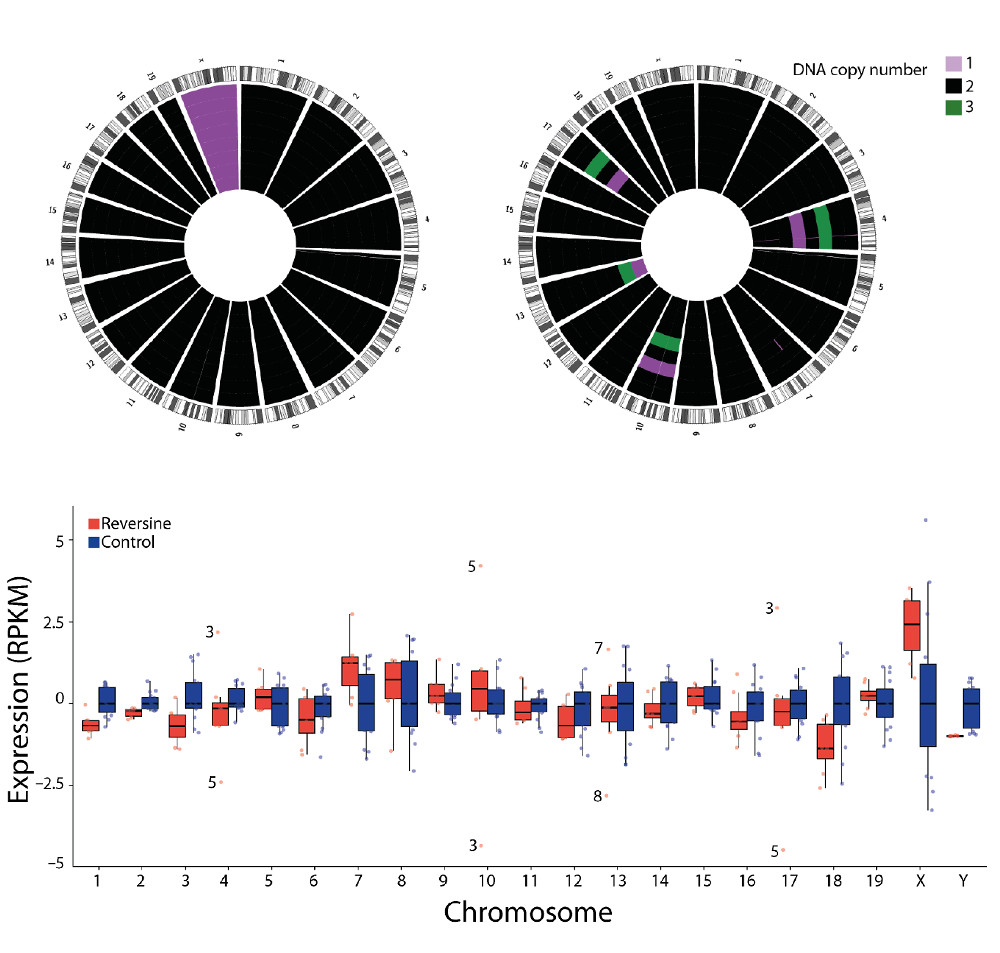
To test the method, researchers sequenced two different cell lines derived from the same person; one from breast cancer and one from normal blood cells. The results from the normal cells showed, as expected, that the vast majority had two copies of each chromosome. However, many of the cancer cells were missing or had gained pieces of chromosomes, and their transcriptomes were significantly different from the normal cells.
The researchers also spotted a previously unobserved cancer-associated chromosomal fusion in the breast cancer cell line. After seeing abnormalities in the RNA data, the transcriptomes of three of the cells were further analysed using Pacific Biosciences long-read sequencing machines. These readings showed the complete sequence of a fusion transcript arising from the fusion of two genes on different chromosomes. Because the genomes of the same single cells were sequenced in parallel, the position where two different chromosomes had fused, generating this new transcript, could be detected.
An unexpected result in the parallel sequence data of the normal blood cell line demonstrates the potential of this method to see detail that was previously invisible. While the data for most cells was similar, four cells were discovered to have three copies of chromosome 11 each; this was reflected in the RNA expression data, which peaks at chromosome 11. With traditional sequencing, these cells would go unnoticed but scientists are now equipped to ask what effect minute changes like these have on the cells.
G&T-seq is currently being used to better understand early embryonic development. In this study, normal early-stage mouse embryos were compared with mouse embryos treated with a chemical that induces chromosomal missegregation. Once again, the method showed that when a cell gains or loses a chromosome, even after a single cell division, changes in gene expression dosage can immediately be seen.
“Using this method, we’ve been able to reveal cellular properties that cannot be seen by DNA or RNA sequencing alone. This kind of integrated analysis, which we hope will soon also include epigenetic data, allows us a more complete understanding of the extent and evolution of cellular heterogeneity in normal development and disease processes.”
Professor Chris Ponting A corresponding author from the University of Oxford and an Associate Faculty Member at the Sanger Institute
More information
Funding
This work was supported by the UK Wellcome Trust and funding from the Belgian Research Foundation Flanders (FWO) and the University of Leuven (KU Leuven). [FWO-G.0687.12]. Researchers were also supported by the UK Medical Research Council, St John’s College, Cambridge, the New Zealand Woolf-Fisher Trust and the EU Seventh Framework Programme (FP7/2007-2013) under grant agreement no. 262055.
Participating Centres
- Wellcome Trust Sanger Institute
- University of Oxford
- University of Leuven
- University of Cambridge
- Stanford University
- New York Genome Center
Publications:
Selected websites
KU Leuven
Situated in Belgium, in the heart of Western Europe, KU Leuven has been a centre of learning for nearly six centuries. Today, it is Belgium’s largest university and, founded in 1425, one of the oldest and most renowned universities in Europe. As a leading European research university and co-founder of the League of European Research Universities, KU Leuven offers a wide variety of international master’s programmes, all supported by high-quality, innovative, interdisciplinary research.
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
The Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.


