Staying one strep ahead
New research provides the first detailed genetic picture of an evolutionary war between Streptococcus pneumoniae bacteria and the vaccines and antibiotics used against it over recent decades. Large-scale genome sequencing reveals patterns of adaptation and the spread of a drug-resistant lineage of the S. pneumoniae bacteria.
The study unmasks the genetic events by which bacteria such as S. pneumoniae respond rapidly to new antibiotics and vaccines. The team suggests that knowing the enemy better could improve infection control measures.
S. pneumoniae is responsible for a broad range of human diseases, including pneumonia, ear infection and bacterial meningitis. Since the 1970s, some forms of the bacteria have gained resistance to many of the antibiotics traditionally used to treat the disease. In 2000 S. pneumoniae was responsible for 15 million cases of invasive disease across the globe. A new vaccine was introduced to the US in 2000 in an attempt to control disease resulting from the most common and drug-resistant forms of the bacteria.
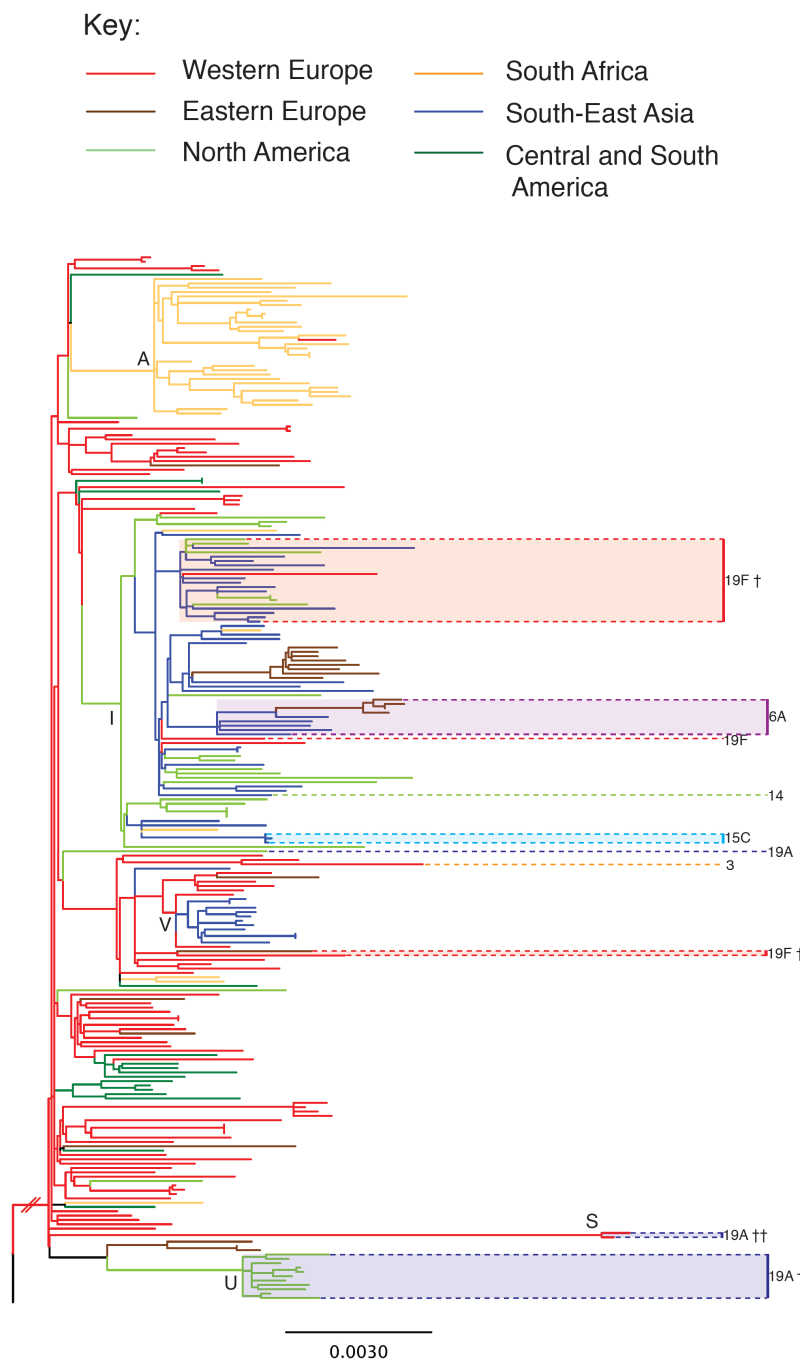
The new research uses DNA sequencing to precisely describe the recent evolution and success of a drug-resistant lineage of the bacteria called PMEN1 that has spread successfully to all continents.
“Drug resistant forms of S. pneumoniae first came onto the radar in the 1970s. We sequenced 240 samples collected over the course of 24 years from the PMEN1 lineage of S. pneumoniae. By comparing the sequences, we can begin to understand how this bacterium evolves and reinvents itself genetically in response to human interventions.”
Dr Stephen Bentley from the Wellcome Trust Sanger Institute and senior author on the study
The power of next-generation sequencing exposes S. pneumoniae as a pathogen that evolves and reinvents itself with remarkable speed. The degree of diversity was a real surprise in such seemingly closely related organisms.
First, the team had to distinguish between single letter mutations that are passed down ‘vertically’ when cells divide in two, and so-called ‘horizontal’ changes – called recombinations – where chunks of DNA letters are passed across from one bacterium to another and swapped over, changing the structure of their genomes.
“Separating these two kinds of change was the critical first step in unlocking the evolutionary history of the PMEN1 lineage. By looking only at the DNA mutations that are passed down through direct ancestry, we constructed an evolutionary tree. When we looked at our tree, we could see that the drug-resistant PMEN1 lineage originated around 1970 – about the time that saw the introduction of the widespread use of antibiotics to fight pneumococcal disease.”
Professor Julian Parkhill Head of Pathogen Genomics at the Wellcome Trust Sanger Institute
The team also use their tree to trace the origin of PMEN1 to Europe, and suggest that the lineage may have been introduced to the Americas and Asia on multiple occasions.
The ‘vertical’ mutations, however, could not fully account for the evolution and adaptability of this pathogen.
The team found that the ‘horizontal’ transfer of DNA had affected three-quarters of the S. pneumoniae genome. The team also found hotspots – areas of the genome that are particularly affected by horizontal transmission.
“We found that genes for antigens – the molecules that trigger our immune response – were particularly prone to this kind of change. The remarkable amount of variation at these hotspots hints at ways S. pneumoniae can evade vaccines against these antigens.
“If the immune system targets these antigens, then the bacteria can simply change them, like a criminal changing their appearance to evade detection.”
Dr William Hanage Associate Professor of Epidemiology at Harvard School of Public Health, and a Visiting Reader at Imperial College London, where he devised the study with scientists at the Wellcome Trust Sanger Institute
The authors also identify differences in the patterns of adaptation in response to antibiotics and vaccines.
“With antibiotics, different strains quite often adapt in the same way to become resistant. With vaccines, it is quite different. What we see is a decline in the prevalence of bacteria that are susceptible to the vaccine. This, in turn, opens the door for bacteria that can evade the vaccine to fill the niche and become the dominant strain.”
Nicholas Croucher from the Wellcome Trust Sanger Institute and first author on the paper
While the latest vaccination measures in the USA have almost completely removed the target pneumococcal strains from the population, the pathogen has deep resources to draw on in response. The research suggests that variants that allowed some bacteria to escape the new vaccine were present before the vaccine was introduced. These variants then flourished, expanding to fill a ‘gap in the market’ as the grip of the dominant strain was weakened through vaccination.
The researchers suggest that the study provides important new clues into the genetic adaptability of bacteria like S. pneumoniae. They suggest that further focused sequencing programs may prove crucial to the future control of this, and other, bacterial pathogens that use similar mechanisms to outsmart human control measures.
More information
Funding
This work was supported by the Wellcome Trust.
Participating Centres
- The Wellcome Trust Sanger Institute, Wellcome Trust Genome campus, Hinxton, Cambridge, UK
- Department of Infectious Disease Epidemiology, Imperial College, St. Mary’s Campus, Norfolk Place, London, UK
- Institute for Medical Microbiology, National Reference Center for Streptococci, University Hospital RWTH Aachen, Germany
- Respiratory Diseases Branch, Centers for Disease Control and Prevention, Atlanta, Georgia, USA
- Respiratory and Meningeal Pathogens Research Unit, National Institute for Communicable Diseases of the National Health Laboratory Service and Univeristy of Witwatersrand, Johannesburg, South Africa
- Samsung Medical Centre, Sungkyunkwan University School of Medicine and Asia Pacific Foundation for Infectious Disease, Seoul, South Korea
- Department of Molecular Cell Biology, Sungkyunkwan University School of Medicine, Suwon, South Korea
- Respiratory and Systemic Infection Laboratory, Health Protection Agency Centre for Infections, London, UK
- The Hospital for Tropical Diseases, Wellcome Trust Major Overseas Programme, Oxford University Clinical Research Unit, Ho Chi Minh City, Vietnam
- Department of Microbiological Surveillance and Research, Statens Serum Institut, Copenhagen, Denmark
- Department of Laboratory Medicine and Pathobiology, University of Toronto and Ontario Agency for Health Protection and Promotion, Ontario, Canada
- Institute of Infection, Immunity and Inflammation, University of Glasgow, Glasgow, UK
- Laboratory of Microbiology, The Rockefeller University, New York, USA
- Hubert Department of Global Health, Rollins School of Public Health and Division of Infectious Diseases, School of Medicine, Emory University, Atlanta, USA
- Department of Epidemiology, Harvard School of Public Health, Boston, MA, USA
Publications:
Selected websites
About Imperial College London
Consistently rated amongst the world’s best universities, Imperial College London is a science-based institution with a reputation for excellence in teaching and research that attracts 14,000 students and 6,000 staff of the highest international quality. Innovative research at the College explores the interface between science, medicine, engineering and business, delivering practical solutions that improve quality of life and the environment – underpinned by a dynamic enterprise culture.
Since its foundation in 1907, Imperial’s contributions to society have included the discovery of penicillin, the development of holography and the foundations of fibre optics. This commitment to the application of research for the benefit of all continues today, with current focuses including interdisciplinary collaborations to improve global health, tackle climate change, develop sustainable sources of energy and address security challenges.
In 2007, Imperial College London and Imperial College Healthcare NHS Trust formed the UK’s first Academic Health Science Centre. This unique partnership aims to improve the quality of life of patients and populations by taking new discoveries and translating them into new therapies as quickly as possible.
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute, which receives the majority of its funding from the Wellcome Trust, was founded in 1992. The Institute is responsible for the completion of the sequence of approximately one-third of the human genome as well as genomes of model organisms and more than 90 pathogen genomes. In October 2006, new funding was awarded by the Wellcome Trust to exploit the wealth of genome data now available to answer important questions about health and disease.
The Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.


