The genetics of heart disease
Two new studies pick out four genome regions that influence the risk of heart disease. The studies, published in Nature Genetics, included more than 20,000 heart disease patients and involved a total of more than 33,000 samples.
The first study, conducted by the Myocardial Infarction Genetics Consortium, examined early-onset heart disease, and found three new genetic loci associated with increased risk. The second, undertaken by the Cardiogenics consortium and involving researchers from more than 30 labs, found a new locus on chromosome 3 strongly associated with heart disease. Previously, only seven regions had been identified.
Although the researchers caution that further work is required, their results suggest that, together, nine of the variants are as good a predictor of Myocardial Infarction (MI) as other established factors such as plasma LDL cholesterol.
Myocardial infarction – commonly known as heart attack – is a leading cause of death worldwide: early-onset MI appears to show some family history and so study of MI in younger people should be valuable in the search for genetic variants involved in increased risk of MI. Both studies started with patients who displayed early-onset MI.
“Our large studies show that much can still be revealed about the genetic causes behind our common disease. Although many decades have been spent searching for genes that influence heart disease, new genetic analyses have the power to pick out three novel SNPs.
“We need to do much more: in larger screens, in looking at more variation and in investigating the roles of the genes we have found, but it is exciting to be bringing these discoveries into the light of further investigation.”
Professor Leena Peltonen Head of Human Genetics at the Wellcome Trust Sanger Institute
Both studies used large initial groups of patients to identify candidate genetic variants – single-letter differences called SNPs – that might be related to MI: they then subjected those candidates to more stringent testing in many more samples. This phased approach is thought to be the most discerning mechanism for finding strong candidates.
The MIGEN study identified, in an independent sample, four regions that had been previously reported by other researchers. Such reconfirmation supports the validity of other findings of the work. The roles of the genes within the three novel regions identified in this work still remain to be determined. There is a suggestion that one gene, PHARCTR1, might lead directly to MI by promoting hardening of arteries.
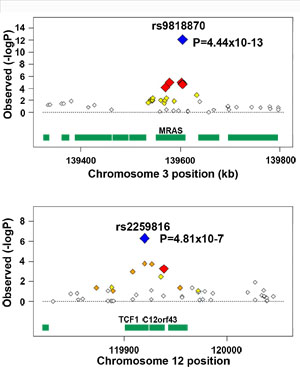
The Cardiogenics consortium, in partnership with the Wellcome Trust Case Control Consortium, extended the initial study which reported six loci associated with Coronary Artery Disease back in 2007, identifying two novel loci strongly associated with risk to this condition. One region on chromosome 3 was refined through more than 25,000 samples to point towards a gene called MRAS that has been shown to be active in the heart.
“We had already found several regions that were implicated with CAD risk, but enlarging the discovery sample was key to identifying new loci. The chromosome 3 region harbouring the MRAS gene stood out as the strongest finding; this is a new confirmed CAD locus. Our work as that of the MIGEN consortium shows that by joining forces we can increase our power to dissect the genetic component of an important common disease such as myocardial infarction.”
Dr Panos Deloukas Senior Investigator at the Wellcome Trust Sanger Institute
Although the effects of the individual variants are modest – at this time the true causative variants remain unknown – the effects of the variants combined were more significant. The risk to individuals in the top 20 per cent for ‘bad’ variants was twice that of those in the bottom 20 per cent.
The results obtained in these studies could be combined with others to examine the usefulness of a test across the best-validated SNPs for increased risk of MI. However, the aggregate genetic studies can explain only a small part of the overall variation in the chance of MI: much remains unclear. The MIGEN study included an examination of the role of structural variants as well as single-base differences in MI, but found no association. Clearly, further studies are required to scan the genome comprehensively for structural variants and epigenetic factors that may contribute to disease risk.
More information
Funding
A full list of participating centres and funding agencies is available at the Nature website.
Publications:
Selected websites
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute, which receives the majority of its funding from the Wellcome Trust, was founded in 1992. The Institute is responsible for the completion of the sequence of approximately one-third of the human genome as well as genomes of model organisms and more than 90 pathogen genomes. In October 2006, new funding was awarded by the Wellcome Trust to exploit the wealth of genome data now available to answer important questions about health and disease.
The Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.


