New drug target for asthma
PD-1 is an existing drug target for some cancers. These findings show that drugs that target PD-1 could also be developed for treating asthma and other inflammatory diseases.
In the first example of using single-cell RNA sequencing to find a therapeutic target, the study also follows how Innate Lymphoid Cells develop. Understanding the biology behind this could help improve PD-1 cancer drugs, which are currently only effective for 20 per cent of patients.
Immune diseases occur when the immune system either fails to remove unwanted cells, such as during infection or cancer, or becomes too active and attacks healthy cells in the body, causing autoimmune diseases or allergies such as asthma.
Innate lymphoid cells (ILC cells) are a recently discovered group of cells in the immune system. Of these, ILC2 cells are important in immune responses against infections and in asthma. The levels of ILC2 cells rise enormously when activated by pollen or toxins, causing lung inflammation, but little is known about how they develop from ILC progenitor cells in the bone marrow or whether there are new specific markers for the activated ones.
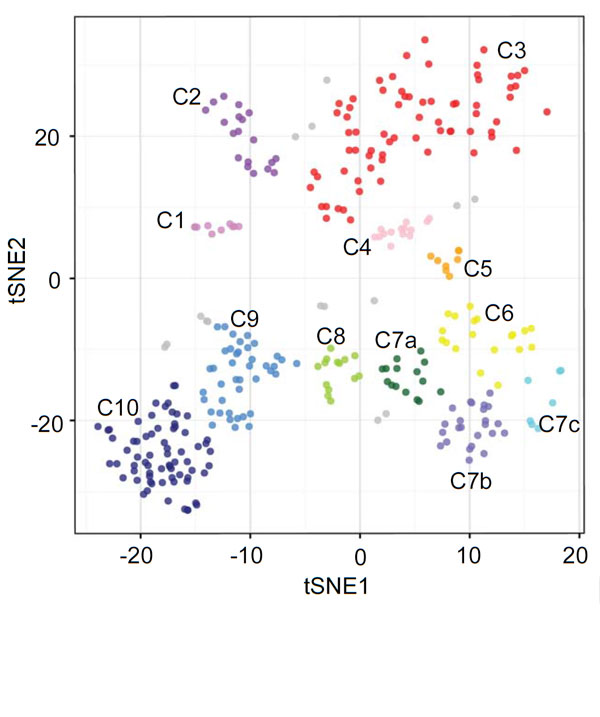
Using Single-Cell RNA sequencing, the researchers studied hundreds of bone marrow cells from mice to investigate how ILCs develop. They were able to clearly identify progenitor cells and different levels of ILC development, and found that the progenitor cells displayed PD-1 protein on their surfaces. Crucially, activated ILC2 cells also displayed high levels of PD-1, which permitted removal of these potentially dangerous cells by a simple antibody treatment targeting PD-1.
“Finding that PD-1 is a marker for the ILC progenitor cells and for activated ILC2s is a really exciting discovery – no-one has seriously looked at this before. It is a very happy coincidence that this interesting gene is already being explored as an immune therapy target for cancer treatment. Antibody drugs targeting PD-1 and thus affecting PD-1-expressing cells may be able to prevent the over-active immune response of ILC2 cells, and reduce or remove asthma symptoms.”
Dr Pentao Liu From the Sanger Institute
PD-1 expression on immune T cells has been previously implicated in the failure of T cells to kill cancer, and therapies with antibodies that target PD-1 on T cells have already been developed for cancers such as melanoma. With the discovery of PD-1 on the ILC cells, scientists hope that these existing drugs and new ones may be able to improve cancer therapy or help asthma sufferers.
“This study helps us understand the biology of the immune system in ways that were impossible previously. If we want to know how to affect the activity of the ILC cells, we need to know how they develop and what switches them on and off. Not only is this useful for asthma and other inflammatory diseases, it could also help us understand what is happening during PD-1 cancer treatment and could potentially make that cancer therapy more effective.”
Dr Yong Yu The first author from the Sanger Institute
“This is a great step forward in our understanding of immune cells. Finding that PD-1 is expressed on activated ILC2 cells gives us an opportunity to manipulate these cells. Further research in this area is needed to help improve immune function as well as prevent immune-mediated inflammatory disease.”
Dr Andrew McKenzie An author on the paper from the MRC Laboratory of Molecular Biology
More information
Funding
This research is funded by the Wellcome Trust, the Medical Research Council, Shanghai Municipal Agricultural Committee, National Natural Science Foundation of China, the Australian Research Council and the National Health and Medical Research Council.
Publications:
Selected websites
MRC Laboratory of Molecular Biology
The Medical Research Council (MRC) Laboratory of Molecular Biology (LMB) is one of the world’s leading research institutes. Discoveries and inventions developed at the LMB, for example DNA sequencing and methods to determine the structure of proteins, have revolutionised all areas of biology. Its scientists work to advance understanding of biological processes at the molecular level. This information will help us to understand the workings of complex systems, such as the immune system and the brain, and solve key problems in human health.
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
Wellcome
Wellcome exists to improve health for everyone by helping great ideas to thrive. We’re a global charitable foundation, both politically and financially independent. We support scientists and researchers, take on big problems, fuel imaginations and spark debate.