Monitoring malaria parasite reveals evolving drug resistance and infection history
Scientists have discovered that the malaria parasite Plasmodium vivax is evolving rapidly to adapt to conditions in different geographical locations, in particular to defend itself against widely-used antimalarial drugs. The study, published in Nature Genetics today (27 June), provides a foundation for using genomic surveillance to guide effective strategies for malaria control and elimination.
P. vivax is mainly found in Asia and South America, and 2.5 billion people are at risk of infection worldwide. This species of malaria parasite is notoriously difficult to work with and the new study has created one of the largest genomic data sets of this species to date, which is available to all researchers.
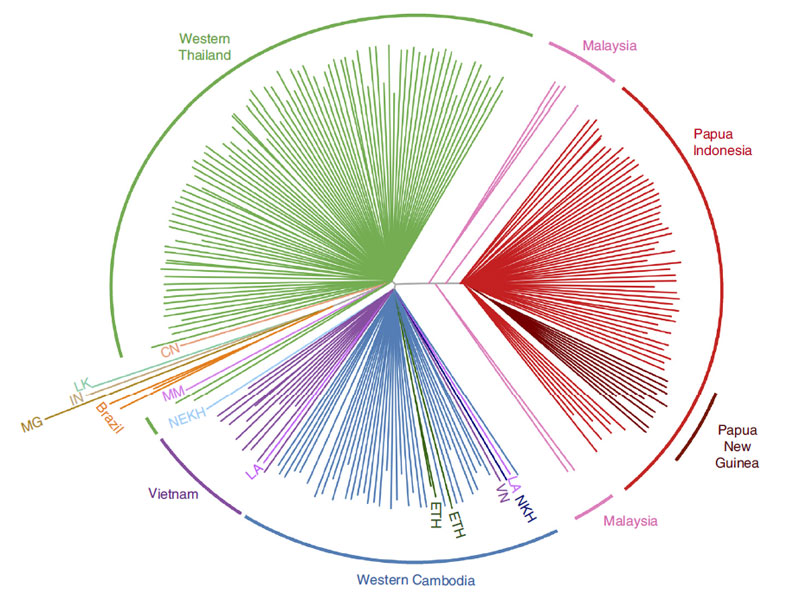
The international team of researchers led by Professor Dominic Kwiatkowski from the Wellcome Trust Sanger Institute and Wellcome Trust Centre for Human Genetics, studied the genomes of more than 200 parasite samples from multiple locations across Southeast Asia, identifying the strains carried by each patient and revealing their infection history.
Unlike Plasmodium falciparum, its more widely studied cousin, Plasmodium vivax can remain dormant inside a person’s liver for years until it emerges causing a malaria relapse. It is extremely difficult to grow under laboratory conditions, and patients have very low levels of P. vivax DNA in their blood. Only now, with modern DNA sequencing technologies, has it been possible to look in detail at the genetics of the parasite that causes relapsing malaria.
The researchers found that the parasites are evolving rapidly to evade anti-malarial drugs. They revealed that P. vivax evolved differently in Thailand, Cambodia and Indonesia, probably due to the different drug use in each place. This study showed that the genomic signals could help identify local areas of drug resistance as they emerged, information that could support local public health staff.
“We can see in the genome that drug resistance is a huge driver for evolution. Intriguingly, in some places, this process appears to be happening in response to drugs used primarily to treat a different malaria parasite, P. falciparum. Although the exact cause isn’t known, this is a worrying sign that drug resistance is becoming deeply entrenched in the parasite population.”
Dr Richard Pearson First author of the paper from the Sanger Institute
“The front line drug used to treat vivax malaria is chloroquine. Our study shows that the strongest evidence of evolution is in Papua, Indonesia, where resistance of P. vivax to chloroquine is now rampant. These data provide crucial information from which we can start to identify the mechanisms of drug resistance in P. vivax.”
Professor Ric Price From the University of Oxford and Menzies School of Health Research, Australia
There are several possible reasons why P. vivax may be evolving to evade drugs used against P. falciparum. Many people carry mixed infections of both species of parasite, so that in treating one species the other automatically gets exposed to the drug. Unsupervised drug use where many people take the most readily available, rather than the most suitable, antimalarial drug may also contribute.A further key finding was that, when the researchers identified patients who were carrying multiple strains of parasite in their blood, the genomic data made it possible to determine how closely the different strains were related to one another.
“This means that we can now start to pull apart the genetic complexity of individual Plasmodium vivax infections, and work out whether the parasites came from one or more mosquito bites. It provides a way of addressing fundamental questions about how P. vivax is transmitted and how it persists within a community, and in particular about the biology of relapsing infections.
“For so long, it’s not been possible to study P. vivax genomes in detail, on a large-scale, but now we can – and we’re seeing the effect that drug use has on how parasites are evolving. In the near future, genomic data will provide more powerful surveillance tools to those who are trying to tackle these problems and make decisions about how to control and eliminate malaria.”
Professor Dominic Kwiatkowski Project leader at Wellcome Trust Sanger Institute and Wellcome Trust Centre for Human Genetics
More information
What is malaria?
- Spread by mosquitos, malaria is one of the most common infectious diseases and a global public health challenge.
- Malaria is a life-threatening disease caused by a parasite that is transmitted through the bite of infected female Anopheles mosquitoes.
- The parasite that causes malaria is a microscopic, single-celled organism called Plasmodium.
- There are six different species of malaria parasite that cause malaria in humans but Plasmodium falciparum and Plasmodium vivax are the most common types.
- Malaria is predominantly found in the tropical and sub-tropical areas of Africa, South America and Asia.
- It is estimated that there were 198 million cases of malaria in 2013 and 584,000 deaths.
- Around 95% of deaths are in children under the age of five living in Sub-Saharan Africa. However, death rates have fallen globally by 47% since 2000.
- If not detected and treated promptly, malaria can be fatal. However, with the right treatment, started early enough, it can be cured.
For more information about malaria please see http://www.yourgenome.org/facts/what-is-malaria
Participating centres
- Wellcome Trust Sanger Institute, Hinxton, Cambridge CB10 1SA, UK
- MRC Centre for Genomics and Global Health, Wellcome Trust Centre for Human Genetics, Oxford OX3 7BN, UK
- Global and Tropical Health Division, Menzies School of Health Research and Charles Darwin University, Darwin, Northern Territories 0811, Australia
- Mahidol‐Oxford Tropical Medicine Research Unit, Mahidol University, Bangkok 10400, Thailand
- National Institute of Allergy and Infectious Diseases, National Institutes of Health, Rockville, Maryland 20852, USA
- National Centre for Parasitology, Entomology, and Malaria Control, Phnom Penh, Cambodia
- Sampov Meas Referral Hospital, Pursat, Cambodia
- Eijkman Institute for Molecular Biology, Jakarta 10430, Indonesia
- Infectious Diseases Society Sabah-Menzies School of Health Research Clinical Research Unit and Queen Elizabeth Hospital Clinical Research Centre, Kota Kinabalu, Sabah, Malaysia
- Oxford University Clinical Research Unit, Ho Chi Minh City, Vietnam
- Papua New Guinea Institute of Medical Research, Madang, Papua New Guinea
- Faculty of Medicine and Health Sciences, Divine Word University, Madang, Papua New Guinea
- Division of Population Health and Immunity, The Walter and Eliza Hall Institute for Medical Research, Parkville, Victoria, Australia
- Department of Medical Biology, University of Melbourne, Parkville, Victoria, Australia
- Department of Parasitology, Institute of Biomedical Sciences, University of São Paulo, São Paulo, Brazil
- Department of Parasitology, Faculty of Medicine, University of Colombo, Sri Lanka
- Institut Pasteur de Madagascar, Antananarivo, Madagascar
- Jiangsu Institute of Parasitic Diseases, Key Laboratory of Parasitic Disease Control and Prevention (Ministry of Health), Jiangsu Provincial Key Laboratory of Parasite Molecular Biology, Wuxi, Jiangsu, People’s Republic of China
- Shoklo Malaria Research Unit, Mae Sot, Tak 63110, Thailand
- Centre for Tropical Medicine and Global Health, Nuffield Department of Clinical Medicine, University of Oxford, OX3 7LJ, UK
Funding
This research was funded by the Wellcome Trust (098051, 090770/Z/09/Z, 091625), the Medical Research Council (G0600718) and the UK Department for International Development (M006212), the Victorian State Government Operational Infrastructure Support and Australian Government NHMRC IRIISS. This study was also supported in part by the Intramural Research Program of the National Institute of Allergy and Infectious Diseases, National Institutes of Health.
Publications:
Selected websites
MalariaGEN
The Malaria Genomic Epidemiology Network (MalariaGEN) is an international community of researchers working to understand how genetic variation in humans, Plasmodium parasites, and Anopheles mosquitoes affects the biology and epidemiology of malaria – and using this knowledge to develop new tools to inform malaria control. The network currently involves researchers in more than 40 malaria-endemic countries with a coordinating centre at Oxford University and the Wellcome Trust Sanger Institute.
Menzies School of Health Research
Menzies School of Health Research aims to improve health outcomes and reduce health inequality for populations in Australia and the Asia-Pacific region, particularly Aboriginal and Torres Strait Islander communities, through excellence and leadership in research, education and capacity development.
Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.