Genomic study of epidemic dysentery reveals how Europe exported a scourge worldwide
The largest genetic study of the bacterium responsible for epidemic dysentery has revealed that the Shigella dysenteriae pathogen, which remains a real scourge in Africa and Asia, probably originated in Europe.
This research, published in the journal Nature Microbiology, also charts the development of the pathogen’s resistance to antibiotics.
Scientists from the Wellcome Trust Sanger Institute, Institut Pasteur in Paris and international collaborators have uncovered hitherto unknown links between the various outbreaks that have occurred through history. One of the worst scourges to afflict humans throughout the 18th and 19th centuries, dysentery was transmitted from one continent to another via migratory movements and military operations.
The bacterium Shigella dysenteriae type 1, causes life-threatening bloody diarrhoea, and has been responsible for thousands of deaths, especially amongst children in the developing world. The last big epidemic in Central America killed 20,000 people between 1969 and 1972. Despite the isolation of S. dysenteriae strains globally, the origin of each dysentery epidemic and the links between them has remained unclear.
To provide a detailed understanding of this disease, a team of scientists led by Professor Nicholas Thomson from the Wellcome Trust Sanger Institute, and senior author Dr François-Xavier Weill from the Institut Pasteur undertook a huge genomic study using high throughput technologies for bacterial genome sequencing and bioinformatics. They analyzed more than 330 strains of S. dysenteriae type 1 isolated between 1915 (in soldiers taking part in the First World War Gallipoli campaign) and 2011. The strains had been collected by 35 international institutes in 66 countries.
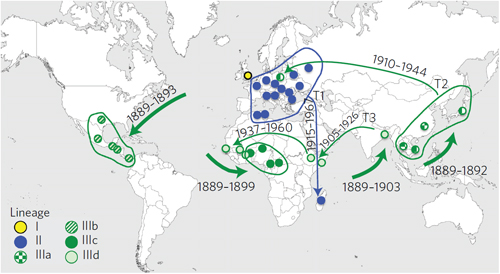
The team found that the type 1 strain has existed since at least the 18th century, and spread throughout the globe. Contrary to popular belief, the study showed that the S. dysenteriae pathogen currently endemic in Africa and Asia is of European origin.
“Analyzing the full genomes of all these Shigella dysenteriae strains collected over a huge timeframe and from such an array of different countries provided us with an unprecedented insight into the historical spread of this pathogen. This was needed because there are still many unanswered questions relating this infamous and important bacterial pathogen. It was achieved by combining high-resolution genomic research data with the detailed information recording the provenance of each sample from a large number of dedicated groups.”
Professor Nicholas Thomson Leader of the Bacterial Genomics and Evolution group at the Sanger Institute
By identifying different genetic lineages, the scientists were able to trace the path of the bacterium worldwide over time, showing that European colonialism and migration helped spread the pathogen.
European S. dysenteriae spread to America, Africa and Asia between 1889 and 1903, aided by European emigration to America and the colonization of territories in Africa and Asia. The bacterium reappeared in Europe during the First and Second World Wars, before dying out in Europe. However, it continued to spread across Asia, Africa and Central America with violent outbreaks, and several epidemic waves then spread to Africa and South-East Asia from the Indian subcontinent.
Since the first bacteria were isolated well before the use of antibiotics, study of the collection has revealed that the first antibiotic resistance appeared in Asia and America in the mid-1960s. The bacterium then acquired resistance genes against most classes of antibiotics – fewer than 1 per cent of bacterial strains have remained susceptible to antibiotics since the 1990s. Scientists consider it inevitable and a cause for concern that dysentery bacteria will acquire resistance to the last-resort antibiotic classes.
“This bacterium is still in circulation, and could be responsible for future epidemics if conditions should prove favorable – such as a large gathering of people without access to drinking water or treatment of human waste. This study highlights the need for an effective vaccine, which will be crucial for controlling this disease in the future in view of the reduced efficacy of antibiotics.”
Dr François-Xavier Weill Research Director at the Enteric Bacterial Pathogens Unit, Institut Pasteur
More information
Funding:
This study received funding from the Institut Pasteur, the Institut Pasteur International Network, InVS, IBEID laboratory of excellence, Le Roch Les Mousquetaires Foundation, and the Wellcome Trust.
Participating Centres:
- Institut Pasteur, Enteric Bacterial Pathogens Unit, 75724 Paris Cedex 15, France.
- Centre for Systems Genomics, University of Melbourne, Parkville, Victoria 3010, Australia.
- Department of Biochemistry and Molecular Biology, Bio21 Molecular Science and Biotechnology Institute, University of Melbourne, Parkville, Victoria 3010, Australia.
- Wellcome Trust Sanger Institute, Cambridge CB10 1SA, UK.
Please see the paper for the full list of funders.
Publications:
Selected websites
Institut Pasteur
The Institut Pasteur, a private foundation with officially recognized charitable status set up by Louis Pasteur in 1887, is today an internationally renowned center for biomedical research with a network of 33 institutes worldwide. In the pursuit of its mission to prevent and fight against diseases in France and throughout the world, the Institut Pasteur operates in four main areas: scientific and medical research, public health and health monitoring, teaching, and business development and technology transfer.
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
The Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.