Methylation linked to metabolic disease
Charting DNA methylation is vital to understanding genetic regions associated with disease. These chemical tags form part of the epigenome: the chemical and structural changes to DNA that affect the function of a person’s genome. The team examined the genomes of fat cells taken from twins and found that DNA methylation patterns are very likely to be passed down from parents to their children.
Twin studies can help to tease out to what extent variation in DNA methylation is genetic and how much is due to environmental factors encountered in a lifetime. For twins that are genetically identical from birth, differences in disease development are likely to be due to environmental influences.
DNA methylation is an epigenetic change but is often under genetic control. The most common form of DNA methylation involves the addition of a molecule, known as a methyl group, to the DNA base cysteine. This process is vital to the regulation of many cellular processes such as embryonic development, transcription, and chromatin structure. Consistent with these important roles, a growing number of human diseases have been associated with aberrant DNA methylation.
“We are exploring epigenetic variation in people, trying to understand the interplay between gene expression and DNA methylation and how they impact disease. Our research has uncovered many links between methylation and genetic regions in fat tissue associated with metabolic diseases.”
Professor Panos Deloukas From the Wellcome Trust Sanger Institute and Queen Mary University London
The team surveyed more than 450,000 sites on the genome from fat tissue samples from 648 twins, the largest twin study of its kind. They found that, in apparently healthy people, there is little variation in methylation patterns. Variability is suppressed in regions that are important for gene regulation such as promoters.
When the team integrated genetic variation and DNA methylation they found 28 per cent of sites to be under genetic control. Overlapping this information with the variants known to regulate gene expression showed that only 6 per cent of these variants regulated both gene expression and DNA methylation.
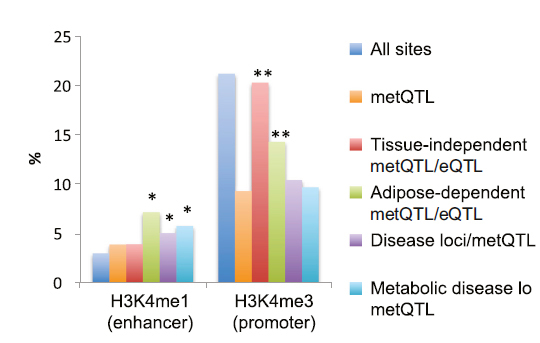
The team integrated the information about DNA methylation with other genetic data from the NIH RoadMap Epigenomics Mapping Consortium, and all known regions in the genome associated with common disease. They found many of the variants that regulate DNA methylation overlap with metabolic-trait or disease loci in regulatory elements known as gene enhancers. The strongest effects were seen for high-density lipoprotein cholesterol and BMI.
“We know that DNA methylation plays a role in gene regulation and disease susceptibility. Through our research we are beginning to unravel the frequency, location and function of DNA methylation variation.”
Elin Grundberg Lead author from the Wellcome Trust Sanger Institute and McGill University, Montreal, Canada
This study provides functional information for genetic regions associated with metabolic disease. The team did further analysis on a variant known to be associated with increased BMI, close to the ADCY3 gene. They found that this variant also regulates DNA methylation in the same region.This study has greatly enhanced the understanding of the link between methylation and disease in specific cell types. The next step for the team is to explore further the effects of methylation on other tissue types such as blood and skin cells and start mapping shared versus cell-restricted sites of DNA methylation.
More information
Funding
A full list of funding can be found on the paper.
Participating Centres
- Wellcome Trust Sanger Institute, CB101SA Hinxton, UK
- Department of Twin Research and Genetic Epidemiology, King’s College London, SE17EH London, UK
- Wellcome Trust Centre for Human Genetics, University of Oxford, OX37BN Oxford, UK
- Department of Genetic Medicine and Development and Institute for Genetics and Genomics in Geneva, University of Geneva Medical School, 1211 Geneva, Switzerland
- Department of Human Genetics, McGill University and Genome Quebec Innovation Centre, Montreal, QC H3A1A5, Canada
- Oxford Centre for Diabetes, Endocrinology, & Metabolism, University of Oxford, Churchill Hospital, OX37LJ Oxford, UK
- NIHR Oxford Biomedical Research Centre, Churchill Hospital, OX3 7LE Oxford, UK
- Molecular Medicine, Department of Medical Sciences, Uppsala University, 751 85 Uppsala, Sweden
- Science for Life Laboratory, Uppsala University, 751 23 Uppsala, Sweden
- William Harvey Research Institute, Barts and The London School of Medicine and Dentistry, Queen Mary University of London, EC1M 6BQ London, UK
Publications:
Selected websites
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
The Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.