Paths to Cardiovascular Diseases and Type 2 Diabetes
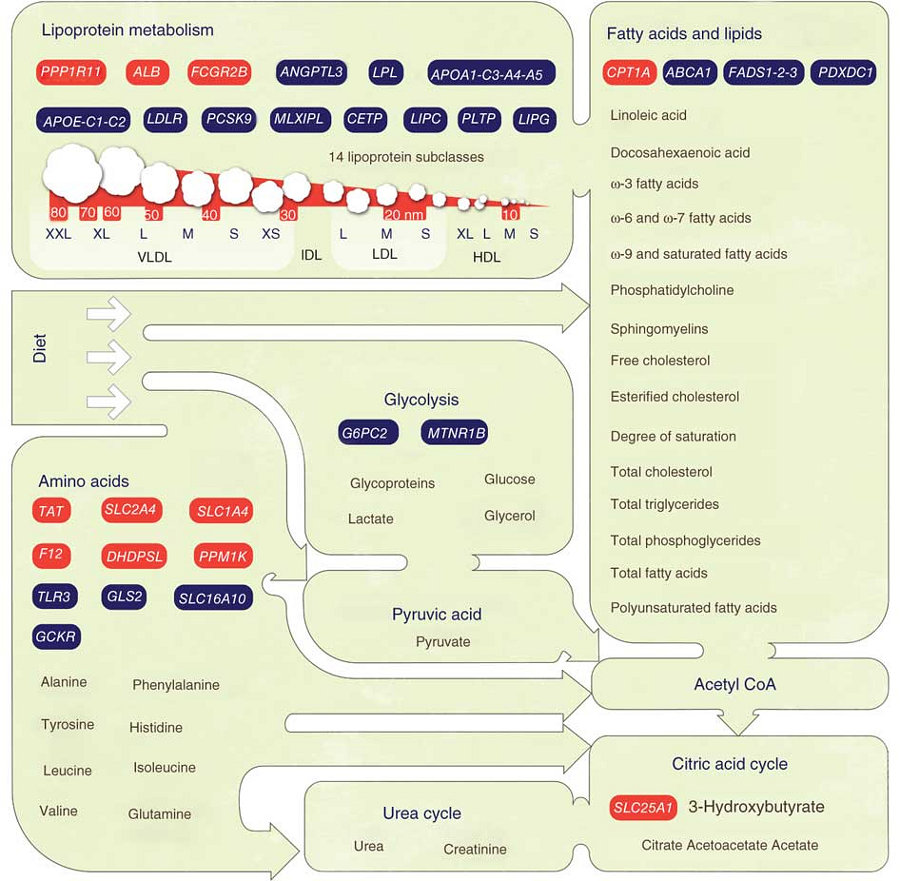
The team studied genetic variation in Finnish population samples and identified 31 regions of the genome that regulate metabolite levels. Many of these metabolites act as biological markers in the body for common disorders. This was the largest study of its kind with 8330 individuals from six population-based cohorts taking part and 7.7 million genomic markers studied.
Metabolites are small molecules that take part in various chemical reactions in the human body. They play a key role in many biological pathways and often contribute to the risk of many diseases, particularly disorders of the metabolic and cardiovascular systems. They can be used to identify a cell population, make a diagnosis, and measure the progress of disease or the effects of treatment. General clinical testing can only monitor a few circulating metabolites.
“We used a high throughput method called nuclear magnetic resonance that can measure more than a hundred different metabolites at a time. This gave us a more in-depth picture of circulating metabolic compounds than can be obtained from routine laboratory tests of metabolism. Using nuclear magnetic resonance technology we were able to identify a large number of genetic regions regulating the level of compounds circulating in the blood stream.”
Dr Nelson Freimer Co-author from UCLA
Of the 31 genetic regions the team found to be associated with levels of the metabolites, 11 had never been associated with any metabolic function. Also, two of the regions were well-established indicators for cardiovascular disease and five of the regions were affecting recently discovered amino acids that are potential biomarkers for type 2 diabetes.
The team found also that the metabolic measures reported here show higher heritability than comparable clinically used serum metabolites. They accomplished this by comparing the metabolite levels in 561 identical and fraternal twin pair. They saw that the identified genetic markers explained a greater component of the heritability than typically reported for clinically used measures. These results suggest that the studied metabolites are describing better the underlying biology than the routinely used laboratory tests.
“Our results highlight the power of large-scale population-based studies to learn about genetic effects of metabolic variation. Our study provides further support for the use of detailed data of a large number of metabolites in genetic studies to provide biological insights and to help to understand the processes leading to these common diseases.”
Dr Samuli Ripatti Lead author and Honorary Faculty Member at the Wellcome Trust Sanger Institute and a group leader at Institute for Molecular Medicine Finland (FIMM)
More information
Funding
Study has been supported by grants from the Academy of Finland, EU FP7 Program, Sigrid Juselius Foundation, Finnish Foundation for Cardiovascular Diseases, The US National Heart, Lung, and Blood Institute and the Wellcome Trust.
Participating Centres
List of participating centres can be found in the paper.
Publications:
Selected websites
The Institute for Molecular Medicine Finland
The Institute for Molecular Medicine Finland (FIMM) is an international research institute focusing on building a bridge from discovery to medical applications. FIMM investigates molecular mechanisms of disease using genomics and medical systems biology in order to promote human health. FIMM is a multi-disciplinary institute combining high-quality science with unique research cohorts and patient materials, and state-of-the-art technologies.
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
The Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.