Low doses of radiation promote cancer-capable cells
New research in mice helps to understand the risks around exposure to low doses of radiation, such as CT scans and x-rays
Low doses of radiation equivalent to three CT scans, which are considered safe, give cancer-capable cells a competitive advantage over normal cells in healthy tissue, scientists have discovered. Researchers at the Wellcome Sanger Institute and the University of Cambridge studied the effects of low doses of radiation in the oesophagus of mice.
The team found that low doses of radiation increase the number of cells with mutations in p53, a well-known genetic change associated with cancer. However, giving the mice an antioxidant before radiation promoted the growth of healthy cells, which outcompeted and replaced the p53 mutant cells.
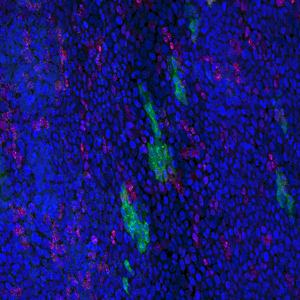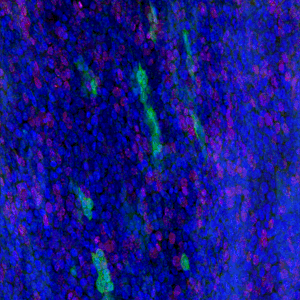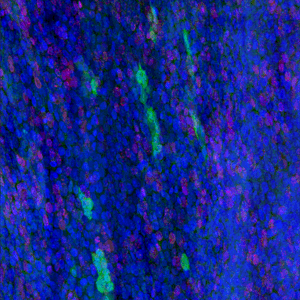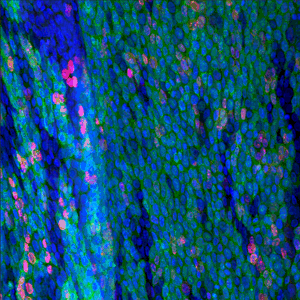
The results, published today (18 July) in Cell Stem Cell show that low doses of radiation promote the spread of cancer-capable cells in healthy tissue. Researchers recommend that this risk should be considered in assessing radiation safety. The study also offers the possibility of developing non-toxic preventative measures to cut the risk of developing cancer by bolstering our healthy cells to outcompete and eradicate cancer-capable cells.
Every day we are exposed to various sources of ionising radiation, including natural radiation in soil and rock, and important medical procedures like CT scans and x-rays.
Low doses of radiation, such as the exposure from medical imaging, are considered safe as they cause little DNA damage and apparently minimal effect on long-term health. Until now, other effects of exposure to low levels of radiation have remained hidden, meaning understanding the true risk associated with low doses of radiation has been difficult.
Researchers have previously shown that our normal tissues, like skin, are battlefields where mutant cells compete for space against healthy cells. We all have cancer-capable mutant cells in healthy tissues, including those with p53 mutations, which increase in number as we age, yet very few eventually go on to form cancer.
In this new study, researchers show that low doses of radiation weigh the odds in favour of cancer-capable mutant cells in the oesophagus. The Sanger Institute researchers and their collaborators gave mice a 50 milligray dose of radiation, equivalent to three or four CT scans. As a result, the p53 mutant cells spread and outcompeted healthy cells.
“Our bodies are the set of ‘Game of Clones’ – a continuous battle for space between normal and mutant cells. We show that even low doses of radiation, similar to three CT scans’ worth, can weigh the odds in favour of cancer-capable mutant cells. We’ve uncovered an additional potential cancer risk as a result of radiation that needs to be recognised.”
Dr David Fernandez-Antoran First author from the Wellcome Sanger Institute
Researchers then gave the mice an over-the-counter antioxidant – N-Acetyl Cysteine (NAC) – before exposure to the same level of radiation. The team discovered that the antioxidant gave normal cells the boost needed to outcompete and eradicate the p53 mutant cells.
However, the antioxidant alone without exposure to radiation did not help normal cells battle the mutant clones.
“Giving mice an antioxidant before exposing them to low doses of radiation gave healthy cells the extra boost needed to fight against the mutant cells in the oesophagus and make them disappear. However, we don’t know the effect this therapy would have in other tissues – it could help cancer-capable cells elsewhere become stronger. What we do know is that long term use of antioxidants alone is not effective in preventing cancer in people, according to other studies.”
Dr Kasumi Murai An author from the Wellcome Sanger Institute
“Medical imaging procedures using radiation, such as CT scans and x-rays, have a very low level of risk – so low that it’s hard to measure. This research is helping us understand more about the effects of low doses of radiation and the risks it may carry. More research is needed to understand the effects in people.”
Professor Phil Jones Lead author from the Wellcome Sanger Institute and MRC Cancer Unit, University of Cambridge
The team suggests this research also highlights the possibility of developing therapies to prevent cancer. By making healthy cells fitter they naturally push out cancer-capable cells, without any toxic side effects for the patient.
More information
Publication:
David Fernandez-Antoran et al. (2019) Outcompeting p53-mutant cells in the normal esophagus by redox manipulation. Cell Stem Cell. DOI: 10.1016/j.stem.2019.06.011
Funding:
This study was supported by the European Union (FP7-Euratom-Fission award 323267), the Medical Research Council, Wellcome (098051 and 296194) and Cancer Research UK (C609/A17257).
Selected websites
About the University of Cambridge
The mission of the University of Cambridge is to contribute to society through the pursuit of education, learning and research at the highest international levels of excellence. To date, 107 affiliates of the University have won the Nobel Prize.
Founded in 1209, the University comprises 31 autonomous Colleges, which admit undergraduates and provide small-group tuition, and 150 departments, faculties and institutions. Cambridge is a global university. Its 19,000 student body includes 3,700 international students from 120 countries. Cambridge researchers collaborate with colleagues worldwide, and the University has established larger-scale partnerships in Asia, Africa and America.
The University sits at the heart of the ‘Cambridge cluster’, which employs 60,000 people and has in excess of £12 billion in turnover generated annually by the 4,700 knowledge-intensive firms in and around the city. The city publishes 341 patents per 100,000 residents. www.cam.ac.uk
The Wellcome Sanger Institute
The Wellcome Sanger Institute is a world leading genomics research centre. We undertake large-scale research that forms the foundations of knowledge in biology and medicine. We are open and collaborative; our data, results, tools and technologies are shared across the globe to advance science. Our ambition is vast – we take on projects that are not possible anywhere else. We use the power of genome sequencing to understand and harness the information in DNA. Funded by Wellcome, we have the freedom and support to push the boundaries of genomics. Our findings are used to improve health and to understand life on Earth. Find out more at www.sanger.ac.uk or follow us on Twitter, Facebook, LinkedIn and on our Blog.
About Wellcome
Wellcome exists to improve health by helping great ideas to thrive. We support researchers, we take on big health challenges, we campaign for better science, and we help everyone get involved with science and health research. We are a politically and financially independent foundation.