Anti-fungal drug increases flu susceptibility
Amphotericin B is an important anti-fungal treatment for people with already suppressed immune systems such as cancer patients and bone marrow transplant patients. Cells and mice treated with Amphotericin B lost the protective effects of IFITM3, a protein critical for protecting against viral infection. This increased their susceptibility to influenza infection.
“This is an important discovery and the consequences for patients on certain anti-fungal treatments should now be investigated. Preventative flu vaccinations, rapid antiviral therapy or alternative anti-fungal treatment could be offered to these patients when at risk of flu infection.”
Professor Paul Kellam Author from the Wellcome Trust Sanger Institute
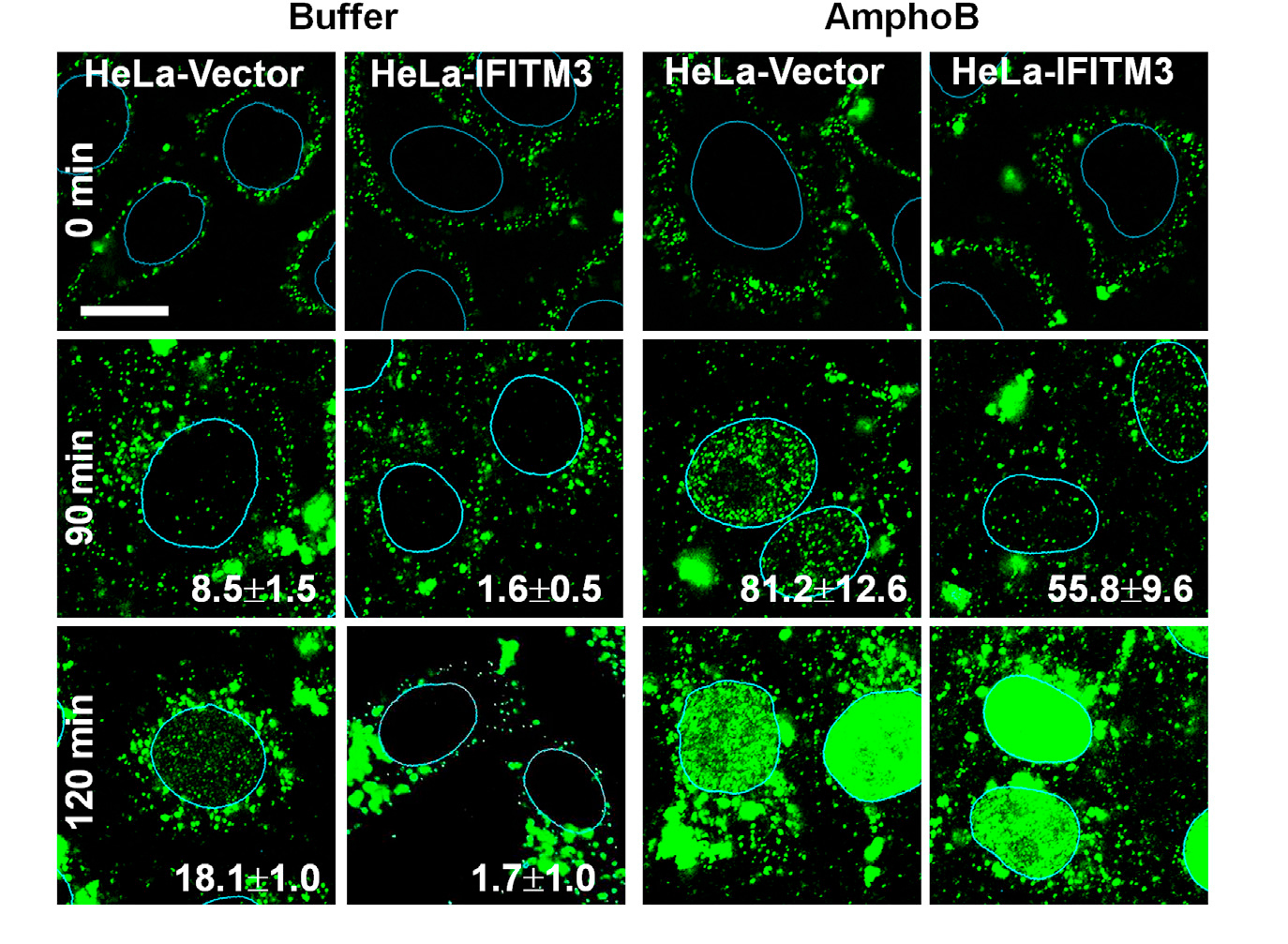
The team discovered that when cell samples were treated with Amphotericin B, the protective effects of IFITM3 were rendered inactive. This allowed the influenza virus to easily infect the cells.
“When we treated lung cancer cells with the anti-fungal drug, we saw the anti-viral protection from IFITM3 pretty much disappear.”
Christopher Chin Co-first author from the University of Massachusetts Medical School
To fully understand the affects Amphotericin B has on IFITM proteins, the team treated mice with the anti-fungal drug. Once the mice contracted influenza, they displayed the same flu symptoms as mice completely lacking the protective IFITM3 gene. In the absence of the influenza virus, the mice treated with the Amphotericin B showed no signs of illness.
Previous studies from the Sanger Institute found that people who have a genetic variant in the IFITM3 gene are more susceptible to severe influenza than people without the variant.
This research indicates that patients undergoing Amphotericin B anti-fungal treatments could potentially lose the protective effects of IFITM3. The loss of IFITM3 could increase the risk of flu infections in patients with already compromised immune systems.
“Sometimes a very useful drug can also have unforeseen effects. We now see that a major part of the body’s natural defences to influenza virus is rendered inactive by Amphotericin B in cells and mice. It’s our hope that reporting the consequences of this interaction may stimulate further translational studies and potentially guide patient care.”
Dr Abraham Brass Senior author in the Microbiology and Physiological Systems Dept. at University of Massachusetts Medical School
The researchers agree that further work is now needed to evaluate if this effect has any clinical significance for patients receiving Amphotericin B-based treatments.
“It’s a fascinating study, and vital that we now work out if these effects in mice are also seen in people. Patients on amphotericin often have complicated medical conditions and may be immune supressed; we need to work out if the treatments these patients were getting for other conditions made their influenza worse, as seen in the mice. If this is the case, we need to vaccinate those at risk, use these treatments sparingly and give antiviral drugs as early as possible when flu is developing. Clinical studies are urgently needed.”
Professor Peter Openshaw Director of the Centre for Respiratory Infections, Imperial College London
This research was part of an ongoing collaboration between University of Massachusetts Medical School and the Wellcome Trust Sanger Institute.
More information
Funding
This work was funded by the Charles H Hood Foundation, the National Institutes of Health, and the Wellcome Trust.
Participating Centres
- Department of Microbiology and Physiological Systems, University of Massachusetts Medical School, Worcester, MA 01655, USA
- Ragon Institute of Massachusetts General Hospital, M.I.T. and Harvard University, Charlestown, MA 02129, USA
- Wellcome Trust Sanger Institute, Wellcome Trust Genome Campus, Hinxton CB10 1SA, UK
- Eidgeno¨ ssische Technische Hochschule, Deutsch English Department of Chemistry and Applied Biosciences, 8093 Zurich, Switzerland
- Department of Genetics, Harvard Medical School, 77 Avenue Louis Pasteur, Boston, MA 02115, USA
- Division of Genetics, Brigham and Women’s Hospital, 77 Avenue Louis Pasteur, Boston, MA 02115, USA
- Howard Hughes Medical Institute, 4000 Jones Bridge Road, Chevy Chase, MD 20815, USA
- MRC/UCL Centre for Medical Molecular Virology, Division of Infection & Immunity, University College London, Gower Street, London W1CE 6BT, UK
Publications:
Selected websites
The Wellcome Trust Sanger Institute
The Wellcome Trust Sanger Institute is one of the world’s leading genome centres. Through its ability to conduct research at scale, it is able to engage in bold and long-term exploratory projects that are designed to influence and empower medical science globally. Institute research findings, generated through its own research programmes and through its leading role in international consortia, are being used to develop new diagnostics and treatments for human disease.
The Wellcome Trust
The Wellcome Trust is a global charitable foundation dedicated to achieving extraordinary improvements in human and animal health. We support the brightest minds in biomedical research and the medical humanities. Our breadth of support includes public engagement, education and the application of research to improve health. We are independent of both political and commercial interests.